. A stream consisting of a mixture of N2 and water vapor, at 98.0 C and 5200.0...
Fantastic news! We've Found the answer you've been seeking!
Question:
 .
.
Transcribed Image Text:
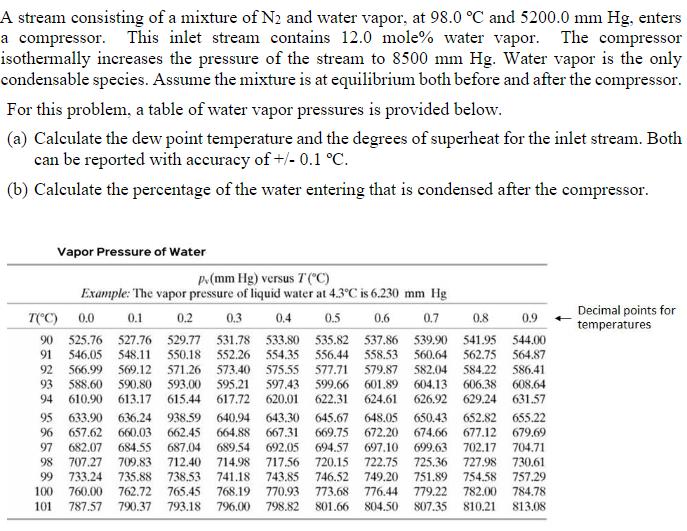
A stream consisting of a mixture of N2 and water vapor, at 98.0 °C and 5200.0 mm Hg, enters a compressor. This inlet stream contains 12.0 mole% water vapor. The compressor isothermally increases the pressure of the stream to 8500 mm Hg. Water vapor is the only condensable species. Assume the mixture is at equilibrium both before and after the compressor. For this problem, a table of water vapor pressures is provided below. (a) Calculate the dew point temperature and the degrees of superheat for the inlet stream. Both can be reported with accuracy of +/- 0.1 °C. (b) Calculate the percentage of the water entering that is condensed after the compressor. Vapor Pressure of Water p.(mm Hg) versus T("C) Example: The vapor pressure of liquid water at 4.3°C is 6.230 mm Hg 0.1 0.3 0.9 Decimal points for temperatures T(*C) 0.0 0.2 0.4 0.5 0.6 0.7 0.8 90 525.76 527.76 529.77 531.78 548,11 533.80 535.82 537.86 539.90 541.95 544.00 91 546.05 550.18 552.26 554.35 556.44 558.53 560.64 562.75 564.87 575.55 597.43 617.72 620.01 582.04 604.13 626.92 629.24 631.57 584.22 586.41 606.38 608.64 92 566.99 569.12 571.26 573.40 577.71 579.87 93 588.60 94 610,90 593.00 615.44 595.21 599.66 622.31 601.89 624,61 590.80 613.17 938.59 662.45 640.94 643.30 667.31 648.05 650.43 674.66 697.10 699.63 725.36 751.89 779.22 652.82 655.22 95 657.62 633.90 636.24 645.67 669.75 96 660.03 664.88 672.20 677.12 679.69 692.05 717.56 702.17 727.98 730.61 754.58 757.29 97 682.07 684.55 687.04 689.54 694.57 704.71 707.27 709,83 735.88 762.72 712.40 714,98 741.18 768,19 98 720,15 722.75 99 733.24 738.53 743.85 746.52 749.20 765.45 770,93 798.82 100 760.00 773.68 776.44 782.00 784.78 101 787.57 790.37 793.18 796.00 801.66 804.50 807.35 810.21 813.08 A stream consisting of a mixture of N2 and water vapor, at 98.0 °C and 5200.0 mm Hg, enters a compressor. This inlet stream contains 12.0 mole% water vapor. The compressor isothermally increases the pressure of the stream to 8500 mm Hg. Water vapor is the only condensable species. Assume the mixture is at equilibrium both before and after the compressor. For this problem, a table of water vapor pressures is provided below. (a) Calculate the dew point temperature and the degrees of superheat for the inlet stream. Both can be reported with accuracy of +/- 0.1 °C. (b) Calculate the percentage of the water entering that is condensed after the compressor. Vapor Pressure of Water p.(mm Hg) versus T("C) Example: The vapor pressure of liquid water at 4.3°C is 6.230 mm Hg 0.1 0.3 0.9 Decimal points for temperatures T(*C) 0.0 0.2 0.4 0.5 0.6 0.7 0.8 90 525.76 527.76 529.77 531.78 548,11 533.80 535.82 537.86 539.90 541.95 544.00 91 546.05 550.18 552.26 554.35 556.44 558.53 560.64 562.75 564.87 575.55 597.43 617.72 620.01 582.04 604.13 626.92 629.24 631.57 584.22 586.41 606.38 608.64 92 566.99 569.12 571.26 573.40 577.71 579.87 93 588.60 94 610,90 593.00 615.44 595.21 599.66 622.31 601.89 624,61 590.80 613.17 938.59 662.45 640.94 643.30 667.31 648.05 650.43 674.66 697.10 699.63 725.36 751.89 779.22 652.82 655.22 95 657.62 633.90 636.24 645.67 669.75 96 660.03 664.88 672.20 677.12 679.69 692.05 717.56 702.17 727.98 730.61 754.58 757.29 97 682.07 684.55 687.04 689.54 694.57 704.71 707.27 709,83 735.88 762.72 712.40 714,98 741.18 768,19 98 720,15 722.75 99 733.24 738.53 743.85 746.52 749.20 765.45 770,93 798.82 100 760.00 773.68 776.44 782.00 784.78 101 787.57 790.37 793.18 796.00 801.66 804.50 807.35 810.21 813.08
Expert Answer:
Answer rating: 100% (QA)
Degree of superheeat is the extra temperature in addition to saturation temperature Part 1 At inlet ... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder,? Ronald W. Rousseau,? Lisa G. Bullard
Posted Date:
Students also viewed these chemical engineering questions
-
An equimolar mixture of carbon dioxide and water vapor at 1 atm and 60oC enter a dehumidifying section where the entire water vapor is condensed and removed from the mixture, and the carbon dioxide...
-
An equimolar mixture of carbon monoxide and water vapor at 1 atm and 298 K enters a reactor operating at steady state. The equilibrium mixture, composed of CO2, CO, H2O(g) and H2, leaves at 2000 K....
-
A stream consisting of 44.6 mole% benzene and 55.4% toluene is fed at a constant rate to a process unit that produces two product streams, one a vapor and the other a liquid. The vapor flow rate is...
-
Find a vector parametrization of the intersection of the surfaces x 2 + y 4 + 2z 3 = 6 and x = y 2 in R 3 .
-
From Exercise 10-7, prepare a schedule of accounts payable and verify that the total of the schedule equals the amount in the controlling account.
-
Consider a market with two securities, one of them risk free with rate of return \(R\), and three future states. Suppose that the risky security has a present value \(S_{1}\) and can take three...
-
The plot below is a FRF taken from impacting and measuring the same point on a structure. It is also known as a driving-point FRF. What can we expect to see in the resulting modal analysis animated...
-
Red Sky Company has gathered the following information. Units in beginning work in process ....... 20,000 Units started into production ........... 72,000 Units in ending work in process ............
-
A baseball catcher is performing a stunt for a television commercial. He will catch a baseball with mass m = 173 g, dropped from rest at a height y = 62 m above his glove. The glove brings the ball...
-
You are about to open your first restaurant. You restaurant will have 125 seats and will be open 5 nights a week. Projected covers per week will average 1200 and you are open 50 weeks out of the...
-
Consider a natural monopoly with a market demand curve of QD=50P. The monopolist's costs are described by TC = 4Q and MC = 4. Marginal revenue is given by MR = 100 - 4Q. (a) Find the quantity that...
-
If all markets were contestable, there would be no need for regulation. Do you agree with this statement? Provide reasons and justifications for your answer.
-
Why is a time lag an important feature of a perfectly contestable market?
-
Why do regulators face a trade-off between competitiveness and the benefit of economies of scale in imperfectly competitive markets?
-
Why is costless exit as important a feature of a perfectly contestable market as costless entry?
-
Why were Leontiefs findings considered paradoxical? Explain.
-
Why are people susceptible to social pressures such as conformity, adopting social roles, and obedience to authority? B) How can a person be more independent in terms of relying on their own thinking?
-
Solve each equation. x 3 - 6x 2 = -8x
-
Nitrogen is bubbled through a liquid mixture that initially contains Equimolar amounts of benzene and toluene. The system pressure is 3 atm and the temperature is 80C. The nitrogen flow rate is 10.0...
-
The little-known rare earth element nauseum (atomic weight = 172) has the interesting property of being completely insoluble in everything but 12-year-old bourbon. This curious fact was discovered in...
-
As part of a design calculation, you must evaluate an enthalpy change for an obscure organic vapor that is to be cooled from 1800C to 150C in a heat exchanger. You search through all the standard...
-
Active-Ingredients is a company supplying baking ingredients in major hypermarkets that are facing intense competition from many local and foreign brands. Active-Ingredients is thinking of using a...
-
Nan-Clean is a newly developed cleaning liquid for milk bottles that uses special enzymes to remove dirt effectively. Nan-Clean has been positioned as a product that is made of natural ingredients...
-
Why do sample surveys have predetermined margins of error?

Study smarter with the SolutionInn App


