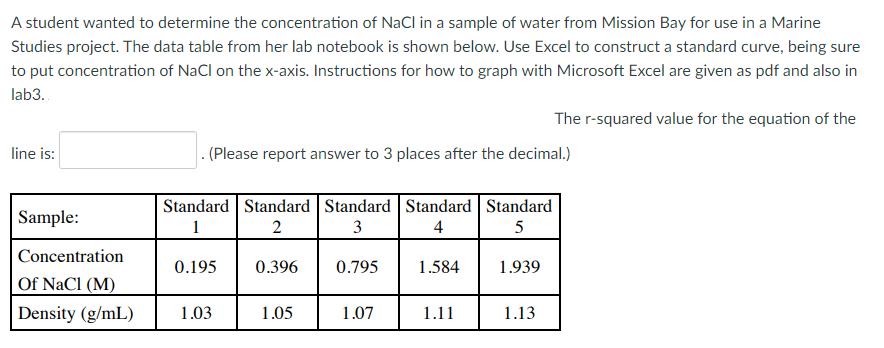
A student wanted to determine the concentration of NaCl in a sample of water from Mission...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A student wanted to determine the concentration of NaCl in a sample of water from Mission Bay for use in a Marine Studies project. The data table from her lab notebook is shown below. Use Excel to construct a standard curve, being sure to put concentration of NaCl on the x-axis. Instructions for how to graph with Microsoft Excel are given as pdf and also in lab3. The r-squared value for the equation of the line is: . (Please report answer to 3 places after the decimal.) Standard Standard Standard Standard Standard Sample: 1 2 3 4 Concentration 0.195 0.396 0.795 1.584 1.939 Of NaCl (M) Density (g/mL) 1.03 1.05 1.07 1.11 1.13 A student wanted to determine the concentration of NaCl in a sample of water from Mission Bay for use in a Marine Studies project. The data table from her lab notebook is shown below. Use Excel to construct a standard curve, being sure to put concentration of NaCl on the x-axis. Instructions for how to graph with Microsoft Excel are given as pdf and also in lab3. The r-squared value for the equation of the line is: . (Please report answer to 3 places after the decimal.) Standard Standard Standard Standard Standard Sample: 1 2 3 4 Concentration 0.195 0.396 0.795 1.584 1.939 Of NaCl (M) Density (g/mL) 1.03 1.05 1.07 1.11 1.13
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A chemist wanted to determine the concentration of a solution of lactic acid, HC3H5O3. She found that the pH of the solution was 2.51. What was the concentration of the solution? The Ka of lactic...
-
A chemist wanted to determine the concentration of a solution of lactic acid, HC 3 H 5 O 3 . She found that the pH of the solution was 2.60. What was the concentration of the solution? The K a of...
-
A chemist wishes to determine the concentration of CrO42- electrochemically. A cell is constructed consisting of a saturated calomel electrode (SCE; see Exercise 24) and a silver wire coated with...
-
The cantilever beam AB shown in the figure is subjected to a concentrated load P at the midpoint and a counterclockwise couple of moment M1 = PL/4 at the free end. Draw the shear-force and...
-
An Erie, Pennsylvania, public indecency ordinance prohibited knowingly or intentionally appearing in public in a state of nudity. Paps, the owners of Kandyland, an Erie establishment featuring...
-
(a) Compute the reactance of a 0.450-H inductor at frequencies of 60.0 Hz and 600 Hz. (b) Compute the reactance of a 2.50-F capacitor at the same frequencies. (c) At what frequency is the reactance...
-
Two electrons move near each other and at the instant shown in Figure P28.85 are \(2.0 \mathrm{~mm}\) apart. The speed of electron 1 is \(v_{1}=300 \mathrm{~m} / \mathrm{s}\), that of electron 2 is...
-
Port Ormond Carpet Company manufactures carpets. Fiber is placed in process in the Spinning Department, where it is spun into yarn. The output of the Spinning Department is transferred to the Tufting...
-
1. Hydrogenation of the above compound in the presence of poisoned palladium catalyst gives Me Me Me H H (a) An optically active compound (c) A racemic mixture (b) An optically inactive compound (d)...
-
Costa, Inc., recently converted from a 5-day, 40-hour workweek to a 4-day, 40-hour workweek, with overtime continuing to be paid at one and one-half times the regular hourly rate for all hours worked...
-
Kebri LTD. sells Radios and Televisions. They have recorded their monthly revenue as follows. Revenue(Y) 330 180 209 390 391 304 347 461 538 610 Radio units (X1) 55 71 92 113 134 156 171 193 219 232...
-
In a 4D Euclidean space with metric ds = dx + dy + dz + dw, we define the 3D surface x + y ++w= R2, with a positive constant R. This surface is called a 3-sphere. We introduce the periodic...
-
How do cognitive biases and perceptual distortions influence the resolution of intricate disputes, and what strategies can be implemented to mitigate their impact ?
-
Carlton Bank has an increase in reserves of $1,000,000. If the reserve ratio is 10%, by what amount may Carlton increase its demand deposits?
-
How can the judicious application of mediation and arbitration principles foster harmonious resolutions amidst intricate intergroup conflicts ?
-
2. Assume that Portugal and England are each able to produce wine and wheat under the following conditions: Portugal England 1 barrel wine 20 labor hrs 1 ton wheat 30 labor hrs 40 labor hrs 30 labor...
-
An American was hitchhiking in Italy and got picked up by an Italian truck driver. The American spoke no Italian, but was fluent in Spanish. He and the truck driver (who spoke no English or Spanish)...
-
Continuation of Exercise 4-83. (a) What is the probability that the first major crack occurs between 12 and 15 miles of the start of inspection? (b) What is the probability that there are no major...
-
Nickel(II) sulfate solution reacts with lithium hydroxide solution to produce a precipitate of nickel(II) hydroxide and a solution of lithium sulfate. Write the molecular equation for this reaction....
-
Use the half-reaction method to balance the equation Ca(s) + Cl2(g) CaCl2(s).
-
Describe the structure of white phosphorus. How does the structure account for the reactivity of this substance?
-
Explain from a technical viewpoint why it is important to distinguish a method, research, approach, and design. Next, briefly discuss how understanding each term individually in addition to how these...
-
Discuss the importance of validity and research design. Next, choose one type of validity (internal, external, construct, or statistical conclusion) and discuss its relevance to experimental, quasi...
-
Define a sampling strategy.

Study smarter with the SolutionInn App


