A student was given an unknown metal. The student determined that the mass of the metal was
Fantastic news! We've Found the answer you've been seeking!
Question:
A student was given an unknown metal. The student determined that the mass of the metal was 30.2 g. The student placed the metal in a graduated cylinder filled with 20.0 mL of water. The metal increased the volume of water to 22.9 mL. Calculate the density of the metal and determine the identity of the metal using the table below. 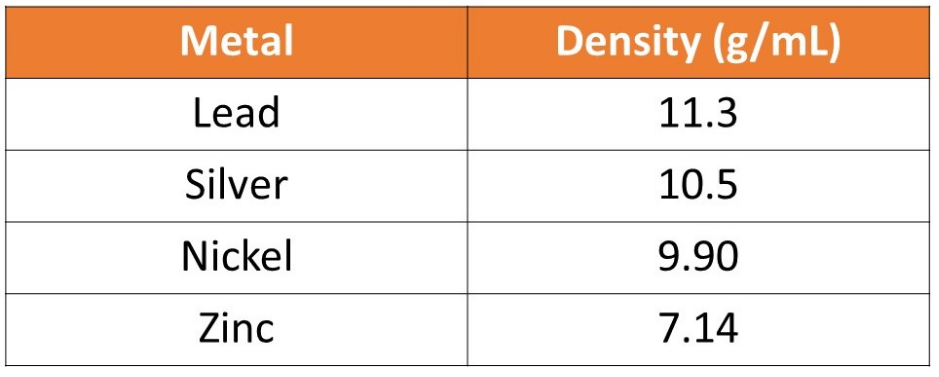
Related Book For 

Fundamentals of Heat and Mass Transfer
ISBN: 978-0471457282
6th Edition
Authors: Incropera, Dewitt, Bergman, Lavine
Posted Date:




