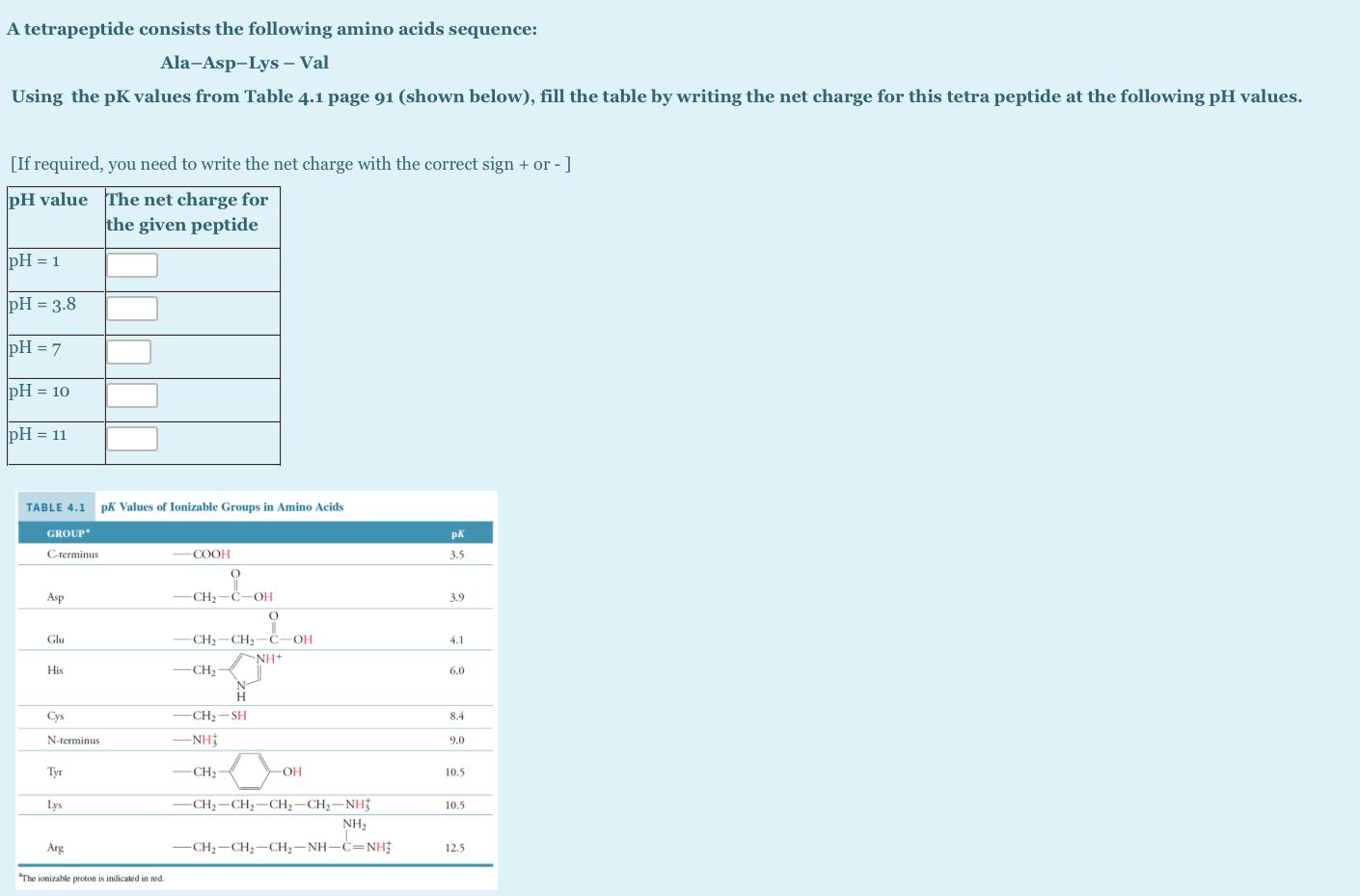
A tetrapeptide consists the following amino acids sequence: Ala-Asp-Lys - Val Using the pK values from...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A tetrapeptide consists the following amino acids sequence: Ala-Asp-Lys - Val Using the pK values from Table 4.1 page 91 (shown below), fill the table by writing the net charge for this tetra peptide at the following pH values. [If required, you need to write the net charge with the correct sign + or -] pH value The net charge for the given peptide pH = 1 pH = 3.8 pH = 7 pH = 10 pH = 11 TABLE 4.1 pK Values of Ionizable Groups in Amino Acids GROUP pK C-rerminus COOH 3.5 Asp CH2-C-OH 3.9 Glu CH2-CH2-C 4.1 NH+ His -CH2 6.0 Cys CH2-SH 8.4 N-terminus -NH 9.0 Tyr -CH2- OH 10.5 Lys -CH2-CH2-CH2-CH2-NH 10.5 NH, Arg -CH;-CH;-CH;-NH-C=NHỊ 12.5 "The ionizable proton is indicated in red A tetrapeptide consists the following amino acids sequence: Ala-Asp-Lys - Val Using the pK values from Table 4.1 page 91 (shown below), fill the table by writing the net charge for this tetra peptide at the following pH values. [If required, you need to write the net charge with the correct sign + or -] pH value The net charge for the given peptide pH = 1 pH = 3.8 pH = 7 pH = 10 pH = 11 TABLE 4.1 pK Values of Ionizable Groups in Amino Acids GROUP pK C-rerminus COOH 3.5 Asp CH2-C-OH 3.9 Glu CH2-CH2-C 4.1 NH+ His -CH2 6.0 Cys CH2-SH 8.4 N-terminus -NH 9.0 Tyr -CH2- OH 10.5 Lys -CH2-CH2-CH2-CH2-NH 10.5 NH, Arg -CH;-CH;-CH;-NH-C=NHỊ 12.5 "The ionizable proton is indicated in red
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In what order would the following amino acids be eluted with a buffer of pH 4 from a column containing an anion-exchange resin? histidine, serine, aspartate, valine
-
You need to write "Observation Report" field research-natural environment about non-verval communication class - What did you looking for - how did you go for looking - what did you find out
-
You need to write a paper about "Shopping Mall Construction Project"
-
Are the marks one receives in a course related to the amount of time spent studying the subject? To analyze this mysterious possibility, a student took a random sample of 10 students who had enrolled...
-
Maves, Inc. booked a deferred tax asset of $ 45,000 due to a basis difference in warranty liabilities. Management has assessed that it is more likely than not that the firm will not realize 30% of...
-
If a monopolist is producing a quantity where marginal revenue is equal to $125 and the marginal cost is equal to $125 which is the price of the good, the monopolist should a. Produce the same...
-
Do neurotransmitters enter the target cell? If not, how do they have an effect on the target cell?
-
You purchase a stock for $100 that pays an annual dividend of $5.50. At the beginning of the second year, you purchase an additional share for $130. At the end of the second year, you sell both...
-
2) On many interstate highways, numbered posts are placed every mile alongside the road. At right is a record of the clock times at which a car passed several mileposts. a) Plot a graph of mile...
-
Kramer Corporation enters a contract to sell 100 custom-made iron pipes to a customer for $300,000 on December 31, 2016, and the customer pays the full amount on that date. The sales contract...
-
Quill Printing allows customers to print their project for a flat fee of $50. However, if a customer prints more than 50 pages, the customer will be charged more in order to cover the costs of the...
-
Draw temperature curves dry air parcels that we release with temperatures of 22C and 24C at the ground. The y-curve in the graph describes the environmental temperature profile. At what altitudes do...
-
Yori Limited issued a dividend of $6.09 yesterday. It expects dividends to grow at a steady rate of 5.63% per annum for many years going forward. If the discount rate for these shares is 11.39% per...
-
If 273.8 years pass, and an isotope of a certain element decays, what percent is left? The half life of the isotope is 122.4 years. Round your answer to the tenths position.
-
Using a normal distribution to model asset prices is a good approximation for low volatilities or short time horizons. What is the weakness of using a normal distribution to model asset prices for...
-
Two small electrified objects A and B are separated by 0.03 m, and repel each other with a force of 4.0x10^-5 N. If we move body A an additional 0.03 m away, what is the electric force now?
-
Problem 2: Aggregate Savings and Labour Supply with a Bequest Motive Consider the following consumer's problem: max U = +1 +2 +y+0+=1, 0
-
Create an appropriate display of the navel data collected in Exercise 25 of Section 3.1. Discuss any special properties of this distribution. Exercise 25 The navel ratio is defined to be a persons...
-
The reaction of hydroxide ion with chloromethane to yield methanol and chloride ion is an example of a general reaction type called a nucleophilic sub- Situation reaction: HO- + CH3C1 CH3OH + C1-...
-
Show the products you would except to obtain by claisen condensation of the following esters: (a) (CH3)2CHCH2COEt (b) Ethyl phenyl acetate (c) Ethyl cyclohexylacetate
-
What product(s) would you expect from Kiliani-Fischer reaction of D-ribose?
-
Which of the seven store departments had the most customer visits, and what was this number?
-
What are the numbers of observations in each of the five categories that rate the respondents level of job satisfaction?
-
What proportion of the 26,280 individuals are in each of the five categories?

Study smarter with the SolutionInn App


