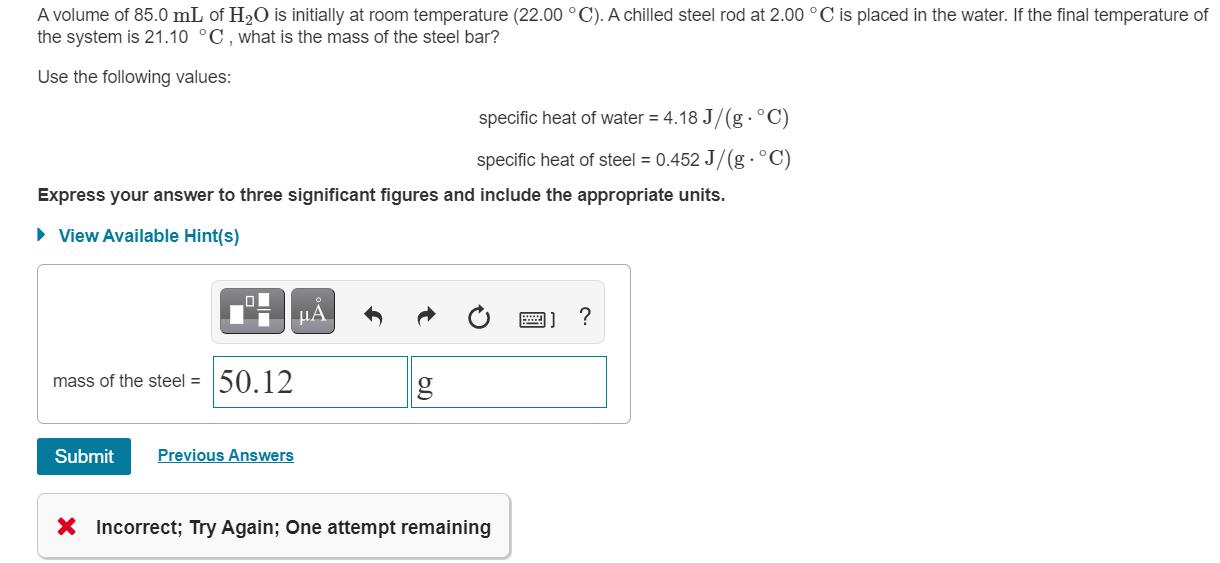
A volume of 85.0 mL of H20 is initially at room temperature (22.00 C). A chilled...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A volume of 85.0 mL of H20 is initially at room temperature (22.00 °C). A chilled steel rod at 2.00 °C is placed in the water. If the final temperature of the system is 21.10 °C, what is the mass of the steel bar? Use the following values: specific heat of water = 4.18 J/(g.° specific heat of steel = 0.452 J/(g.°C) Express your answer to three significant figures and include the appropriate units. • View Available Hint(s) mass of the steel = 50.12 Submit Previous Answers X Incorrect; Try Again; One attempt remaining A volume of 85.0 mL of H20 is initially at room temperature (22.00 °C). A chilled steel rod at 2.00 °C is placed in the water. If the final temperature of the system is 21.10 °C, what is the mass of the steel bar? Use the following values: specific heat of water = 4.18 J/(g.° specific heat of steel = 0.452 J/(g.°C) Express your answer to three significant figures and include the appropriate units. • View Available Hint(s) mass of the steel = 50.12 Submit Previous Answers X Incorrect; Try Again; One attempt remaining
Expert Answer:
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
If a helium-filled balloon initially at room temperature is placed in a freezer, will its volume increase, decrease, or remain the same?
-
If a helium-filled balloon initially at room temperature is placed in a freezer, will its volume increase, decrease, or remain the same?
-
Evaluate each of the following to three significant figures and express each answer in SI units using an appropriate prefix: (a) 354 mg (45 km)/ (0.035 6 k N), (b) (.004 53 Mg) (201 ms), (c) 435 MN/...
-
11. What is the Specific Gravity of Zinc ? 12. The barometric pressure for the day is 14.7 psi. The hangar shop air tank gage reads 120 psi. What is the absolute pressure inside the shop air tank? =...
-
The average selling time for houses sold last year in the greater St. Louis area and the average selling time for houses sold in the area this year appear to be about the same, but the same may not...
-
State your a priori hypothesis about the sign of the slope. Is it reasonable to suppose a cause- and-effect relationship?
-
The price of a share is \(\$ 40\). If \(\mu=0.1\) and \(\sigma^{2}=0.16\) per year, find a \(95 \%\) confidence interval for the price of the share after six months (i.e. an interval...
-
Hart Enterprises recently paid a dividend, D0, of $1.25. It expects to have non-constant growth of 20% for 2 years followed by a constant rate of 5% thereafter. The firms required return is 10%. a....
-
How do organizational learning theories, such as the learning organization concept and knowledge management practices, contribute to sustained competitive advantage and organizational resilience in...
-
During 2020, Richard and Greta Van Fleet, who are married and have two dependent children, ages 14 and 16, and have the following information: Total salaries Bank account interest State of Idaho bond...
-
Identify the oxidation numbers of each element in following molecules.
-
Pennsylvanias Department of Community and Economic Development maintains a network of industrial resource centers (IRCs) that are operated as nonprofit corporations managed by the private sector but...
-
Reliance Control Systems is a large manufacturer of control systems for the electrical power generation and distribution industry. Over the past few years, Reliance has watched overseas competitors...
-
The plant managers of ABC Corporations five plants have met at a common location at 8:00 am every Monday morning for 20 years. The people serving as plant managers have changed several times during...
-
The Quality Institute (TQI) is a partnership of Northwest Florida State College, the University of West Florida, and the Economic Development Council of Okaloosa County. TQI is dedicated to the...
-
Perhaps the most famous case illustrating the enormous cost of winning back lost customers is that of the Tylenol Murders. Seven people in the Chicago area died suddenly after taking Tylenol...
-
I have an RStudio problem where I downloaded Google Stock Pricedata that looks as such:My current R-Code looks like this:# Load libraries library(ggplot2)library(dplyr) library(tidyverse) # Read...
-
Sundial Technologies produces and sells customized network systems in New Brunswick. The company offers a 60-day, all software and labor-and an extra 90-day, parts-only- warranty on all of its...
-
Solid carbon at 25C is burned with a stoichiometric amount of air which is at 1 atm pressure and 25C. Determine the number of moles of CO2 formed per kmol of carbon when only CO2, CO, O2, and N2 are...
-
Submarines change their depth by adding or removing air from rigid ballast tanks, thereby displacing seawater in the tanks. Consider a submarine that has a 700 m3 air-ballast tank originally...
-
Nitrogen enters a converging-diverging nozzle at 700 kPa and 400 K with a negligible velocity. Determine the critical velocity, pressure, temperature, and density in the nozzle.
-
Define a closely held corporation.
-
Explain the role played by a board of directors of a corporation.
-
Explain the steps in forming a limited liability corporation.

Study smarter with the SolutionInn App



