A water sample (50 ml in volume) was used to determine its alkalinity; it was titrated...
Fantastic news! We've Found the answer you've been seeking!
Question:
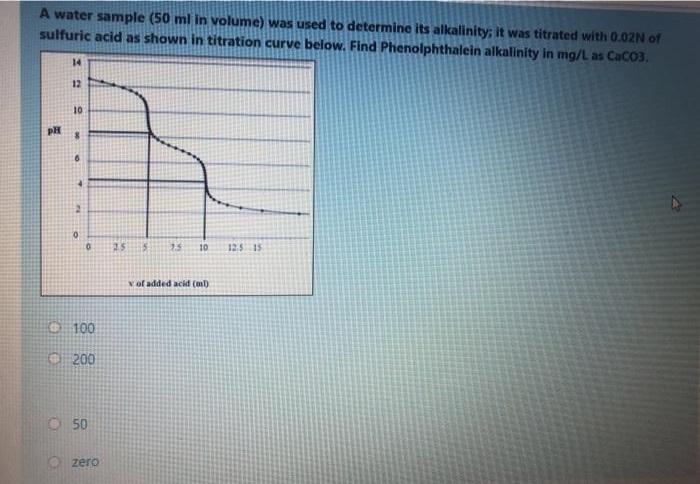
Transcribed Image Text:
A water sample (50 ml in volume) was used to determine its alkalinity; it was titrated with 0.02N of sulfuric acid as shown in titration curve below. Find Phenolphthalein alkalinity in mg/L as CaCO3, 14 12 10 PH 10 125 15 vof added acid (m) 100 O 200 O50 O zero A water sample (50 ml in volume) was used to determine its alkalinity; it was titrated with 0.02N of sulfuric acid as shown in titration curve below. Find Phenolphthalein alkalinity in mg/L as CaCO3, 14 12 10 PH 10 125 15 vof added acid (m) 100 O 200 O50 O zero
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A Knudsen cell was used to determine the vapour pressure of germanium at 1000C. During an interval of7200 s the mass loss through a hole of radius 0.50 mm amounted to 43 ug, what is the vapour...
-
The method of standard additions was used to determine nitrite in a soil sample. A 1.00-mL portion of the sample was mixed with 24.00 mL of a colorimetric reagent, and the nitrite was converted to a...
-
The procedure described in Problem 14-8 was used to determine pH with methyl orange as the indicator. The concentration ratio of the acid to base form of the indicator was 1.84. Calculate the pH of...
-
A curve has equation y = x 2 ln 3x. Find the value of dy/dx and d 2 y/dx2 at the point where x = 2.
-
You've been asked to determine the percentage of students who would support gay marriage. You want to take a random sample of fellow students to make the estimate. Explain whether each of the...
-
Young screenwriter Carl Draper has just finished his first script. It has action, drama, and humor, and he thinks it will be a blockbuster. He takes the script to every motion picture studio in town...
-
Mr. A. Gaylord manages a pension fund and believes that his stock selection ability is excellent. However, he is worried because the market could go down. He considers entering an equity swap where...
-
The income statement of Jones Company for the year ended December 31, 2010, follows. Required a. Compute the net earnings remaining after removing nonrecurring items. b. Determine the earnings (loss)...
-
a) Consider the following proof of logical equivalence: Vx(P(x) Q(x)) = x(P(x) VQ(x)) =Vx(P(x) AQ(x)) = x(P(x) AQ(x))) =(3xP(x) xQ(x)) =(3xP(x)) V (x(Q(x))) =(3xP(x)) V (VxQ(x)) =(3xP(x)) > (VxQ(x))...
-
What are your opinions on this cry closet and on college students' overall workload and responsibilities? Should colleges be addressing the stress students feel more often and/or in a better way? In...
-
Extending the Concepts and Skills 5.85 Roulette. A success, s, in Bernoulli trials is often derived from a collection of outcomes. For example, an American roulette wheel consists of 38 numbers, of...
-
A bullet fired straight through a board \(0.10 \mathrm{~m}\) thick strikes the board with a speed of \(480 \mathrm{~m} / \mathrm{s}\), has constant acceleration through the board, and emerges with a...
-
True or False: The Modified Accelerated Cost Recovery System is the most popular depreciation method used in computing corporate income tax liabilities.
-
A lumber company purchases and installs a wood chipper for \(\$ 200,000\). The chipper is classified as MACRS 7-year property. Its useful life is 10 years. The estimated salvage value at the end of...
-
An object moves from point \(\mathrm{P}\) at \(x=+2.3 \mathrm{~m}\) to point \(\mathrm{Q}\) at \(x=+4.1 \mathrm{~m}\) and then to point \(\mathrm{R}\) at \(x=+1.5 \mathrm{~m}\). (a) What is the \(x\)...
-
The concept similar to depreciation that is applied to natural resources is called what? a. Depletion b. Declining balance c. Amortization d. MACRS
-
The marketing manager of a supermarket chain is trying to see if shelf space can predict sales for breakfast cereal. A random sample of 12 equal-sized stores is selected with the following results....
-
Assume that a trial balance is prepared with an account balance of $21,360 listed as $21,630 and an account balance of $1,500 listed as $15,000. Identify the transposition and the slide.
-
The protein haemerythrin is responsible for binding and carrying 0, in some invertebrates. Each protein molecule has two Fe'+ ions that are in very close proximity and work together to bind one...
-
The cycle involved in the operation of an internal combustion engine is called the Otto cycle. Air can be considered to be the working substance and can be assumed to be a perfect gas. The cycle...
-
The pure rotational microwave spectrum of HCI has absorption lines at the following wave numbers (in cm-1): 21.19, 42.37, 63.56, 84.75, 105.93, 127.12148.31169.49,190.68,211.87,233.06,254.24, 275.43,...
-
At December 31, 2025, the fair value of non-trading securities is 41,300 and the cost is 39,800. At January 1, 2025, there was a credit balance of 900 in the Fair Value Adjustment Non-Trading...
-
On January 1, 2025, Lennon Enterprises acquires 100% of Ono Ltd. for 220,000 in cash. The condensed statements of financial position of the two companies immediately following the acquisition are as...
-
Which of the following will not be reported in the statement of cash flows? a. The net change in plant assets during the year. b. Cash payments for plant assets during the year. c. Cash receipts from...

Study smarter with the SolutionInn App


