Given the time-temperature transformation diagram on page 5, what would be the phases present for a...
Fantastic news! We've Found the answer you've been seeking!
Question:

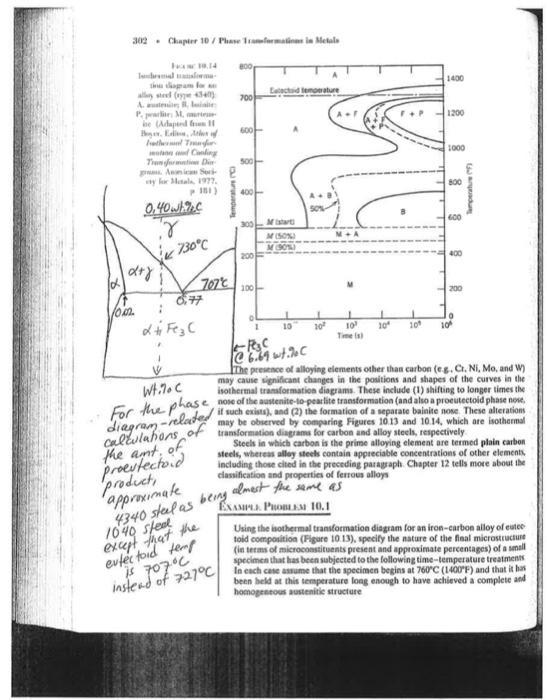
Transcribed Image Text:
Given the time-temperature transformation diagram on page 5, what would be the phases present for a 4340 steel with no alloying elements? Use the bottom of this page to show any work, including any phase diagram-related calculations (worth 5 pts.). A) cooled in air at 2°C/minute to 650°C, then quenched to room temperature B) quenched in cold water directly to room temperature in 0.1 second C) quenched in cold water directly to room temperature in 0.1 second, then heated to 425°C and held there for 1000 seconds, and finally quenched to room temperature D) quenched in cold water to 650°C in 0.1 second, then held at 650°C for 1000 seconds, then quenched to room temperature E) quenched in cold water to 600°C in 0.1 second, then held at 600°C for 5 seconds, then quenched to 350°C, held at 350°C for 60 seconds, and finally quenched to room temperature F) quenched in cold water to 550°C in 0.1 second, then held at 550°C for 5 seconds, then quenched to room temperature G) quenched in cold water to 380°C in 0.1 second, then held at 380°C for 100 seconds, then quenched to room temperature Necessary Steel Phase Diagram Calculations (continued on p. 6 if necessary): 302 Chapter 10/Phase Tranformations in Metals t 10.02. Tural alor diagram for ally steel (ype 4340) A. austen, lie P. pearlie: Mart be (Adapted from I Boyer, Edis, Atherof Fathermal Tr and Cooling Tranformation Di Annican Soci ty for Metals, 1977. p 181) d+y 19.14 0,40whic r 730°C di Fez C 6.77 707°C O aradway is 707°C instead of 727°C/ 800 700 600 500 400- 300 200 100- of 1 Electd temperature Mistart M (50%) 10 ·Pesc @6.69 wt.20 C 10⁰ 10 Time (s) 10 10⁰ 1400 1200 1000 800 600 400 200 10⁰ Temperature (°F) Wt.7. C The presence of alloying elements other than carbon (e.g. Cr. Ni, Mo, and W) may cause significant changes in the positions and shapes of the curves in the isothermal transformation diagrams. These include (1) shifting to longer times the nose of the austenite-to-pearlite transformation (and also a proeutectoid phase nose, it For the phase of diagram-related may be observed by comparing Fig of a separate bainite nose. These alterations calculations the art of proeutectoid product, approximate 4340 steel as being 1040 steel except that the evtec toid temp Figures 10.13 10.14, which are isothermal transformation diagrams for carbon and alloy steels, respectively Steels in which carbon is the prime alloying element are termed plain carbon steels, whereas alloy steels contain appreciable concentrations of other elements, including those cited in the preceding paragraph. Chapter 12 tells more about the classification and properties of ferrous alloys almest the same as EXAMPLE PROBLEM 10.1 Using the isothermal transformation diagram for an iron-carbon alloy of eutee toid composition (Figure 10.13), specify the nature of the final microstructure (in terms of microconstituents present and approximate percentages) of a small specimen that has been subjected to the following time-temperature treatments In each case assume that the specimen begins at 760°C (1400°F) and that it has been held at this temperature long enough to have achieved a complete and homogeneous austenitic structure Temperature (°C) 1700 1500 1300 1100 900 C- 700 500 14 $ 0 4.5% (T) Cu 20 40 Composition (wt% Cu) L TICU Tu 60 Given the time-temperature transformation diagram on page 5, what would be the phases present for a 4340 steel with no alloying elements? Use the bottom of this page to show any work, including any phase diagram-related calculations (worth 5 pts.). A) cooled in air at 2°C/minute to 650°C, then quenched to room temperature B) quenched in cold water directly to room temperature in 0.1 second C) quenched in cold water directly to room temperature in 0.1 second, then heated to 425°C and held there for 1000 seconds, and finally quenched to room temperature D) quenched in cold water to 650°C in 0.1 second, then held at 650°C for 1000 seconds, then quenched to room temperature E) quenched in cold water to 600°C in 0.1 second, then held at 600°C for 5 seconds, then quenched to 350°C, held at 350°C for 60 seconds, and finally quenched to room temperature F) quenched in cold water to 550°C in 0.1 second, then held at 550°C for 5 seconds, then quenched to room temperature G) quenched in cold water to 380°C in 0.1 second, then held at 380°C for 100 seconds, then quenched to room temperature Necessary Steel Phase Diagram Calculations (continued on p. 6 if necessary): 302 Chapter 10/Phase Tranformations in Metals t 10.02. Tural alor diagram for ally steel (ype 4340) A. austen, lie P. pearlie: Mart be (Adapted from I Boyer, Edis, Atherof Fathermal Tr and Cooling Tranformation Di Annican Soci ty for Metals, 1977. p 181) d+y 19.14 0,40whic r 730°C di Fez C 6.77 707°C O aradway is 707°C instead of 727°C/ 800 700 600 500 400- 300 200 100- of 1 Electd temperature Mistart M (50%) 10 ·Pesc @6.69 wt.20 C 10⁰ 10 Time (s) 10 10⁰ 1400 1200 1000 800 600 400 200 10⁰ Temperature (°F) Wt.7. C The presence of alloying elements other than carbon (e.g. Cr. Ni, Mo, and W) may cause significant changes in the positions and shapes of the curves in the isothermal transformation diagrams. These include (1) shifting to longer times the nose of the austenite-to-pearlite transformation (and also a proeutectoid phase nose, it For the phase of diagram-related may be observed by comparing Fig of a separate bainite nose. These alterations calculations the art of proeutectoid product, approximate 4340 steel as being 1040 steel except that the evtec toid temp Figures 10.13 10.14, which are isothermal transformation diagrams for carbon and alloy steels, respectively Steels in which carbon is the prime alloying element are termed plain carbon steels, whereas alloy steels contain appreciable concentrations of other elements, including those cited in the preceding paragraph. Chapter 12 tells more about the classification and properties of ferrous alloys almest the same as EXAMPLE PROBLEM 10.1 Using the isothermal transformation diagram for an iron-carbon alloy of eutee toid composition (Figure 10.13), specify the nature of the final microstructure (in terms of microconstituents present and approximate percentages) of a small specimen that has been subjected to the following time-temperature treatments In each case assume that the specimen begins at 760°C (1400°F) and that it has been held at this temperature long enough to have achieved a complete and homogeneous austenitic structure Temperature (°C) 1700 1500 1300 1100 900 C- 700 500 14 $ 0 4.5% (T) Cu 20 40 Composition (wt% Cu) L TICU Tu 60 Given the time-temperature transformation diagram on page 5, what would be the phases present for a 4340 steel with no alloying elements? Use the bottom of this page to show any work, including any phase diagram-related calculations (worth 5 pts.). A) cooled in air at 2°C/minute to 650°C, then quenched to room temperature B) quenched in cold water directly to room temperature in 0.1 second C) quenched in cold water directly to room temperature in 0.1 second, then heated to 425°C and held there for 1000 seconds, and finally quenched to room temperature D) quenched in cold water to 650°C in 0.1 second, then held at 650°C for 1000 seconds, then quenched to room temperature E) quenched in cold water to 600°C in 0.1 second, then held at 600°C for 5 seconds, then quenched to 350°C, held at 350°C for 60 seconds, and finally quenched to room temperature F) quenched in cold water to 550°C in 0.1 second, then held at 550°C for 5 seconds, then quenched to room temperature G) quenched in cold water to 380°C in 0.1 second, then held at 380°C for 100 seconds, then quenched to room temperature Necessary Steel Phase Diagram Calculations (continued on p. 6 if necessary): 302 Chapter 10/Phase Tranformations in Metals t 10.02. Tural alor diagram for ally steel (ype 4340) A. austen, lie P. pearlie: Mart be (Adapted from I Boyer, Edis, Atherof Fathermal Tr and Cooling Tranformation Di Annican Soci ty for Metals, 1977. p 181) d+y 19.14 0,40whic r 730°C di Fez C 6.77 707°C O aradway is 707°C instead of 727°C/ 800 700 600 500 400- 300 200 100- of 1 Electd temperature Mistart M (50%) 10 ·Pesc @6.69 wt.20 C 10⁰ 10 Time (s) 10 10⁰ 1400 1200 1000 800 600 400 200 10⁰ Temperature (°F) Wt.7. C The presence of alloying elements other than carbon (e.g. Cr. Ni, Mo, and W) may cause significant changes in the positions and shapes of the curves in the isothermal transformation diagrams. These include (1) shifting to longer times the nose of the austenite-to-pearlite transformation (and also a proeutectoid phase nose, it For the phase of diagram-related may be observed by comparing Fig of a separate bainite nose. These alterations calculations the art of proeutectoid product, approximate 4340 steel as being 1040 steel except that the evtec toid temp Figures 10.13 10.14, which are isothermal transformation diagrams for carbon and alloy steels, respectively Steels in which carbon is the prime alloying element are termed plain carbon steels, whereas alloy steels contain appreciable concentrations of other elements, including those cited in the preceding paragraph. Chapter 12 tells more about the classification and properties of ferrous alloys almest the same as EXAMPLE PROBLEM 10.1 Using the isothermal transformation diagram for an iron-carbon alloy of eutee toid composition (Figure 10.13), specify the nature of the final microstructure (in terms of microconstituents present and approximate percentages) of a small specimen that has been subjected to the following time-temperature treatments In each case assume that the specimen begins at 760°C (1400°F) and that it has been held at this temperature long enough to have achieved a complete and homogeneous austenitic structure Temperature (°C) 1700 1500 1300 1100 900 C- 700 500 14 $ 0 4.5% (T) Cu 20 40 Composition (wt% Cu) L TICU Tu 60 Given the time-temperature transformation diagram on page 5, what would be the phases present for a 4340 steel with no alloying elements? Use the bottom of this page to show any work, including any phase diagram-related calculations (worth 5 pts.). A) cooled in air at 2°C/minute to 650°C, then quenched to room temperature B) quenched in cold water directly to room temperature in 0.1 second C) quenched in cold water directly to room temperature in 0.1 second, then heated to 425°C and held there for 1000 seconds, and finally quenched to room temperature D) quenched in cold water to 650°C in 0.1 second, then held at 650°C for 1000 seconds, then quenched to room temperature E) quenched in cold water to 600°C in 0.1 second, then held at 600°C for 5 seconds, then quenched to 350°C, held at 350°C for 60 seconds, and finally quenched to room temperature F) quenched in cold water to 550°C in 0.1 second, then held at 550°C for 5 seconds, then quenched to room temperature G) quenched in cold water to 380°C in 0.1 second, then held at 380°C for 100 seconds, then quenched to room temperature Necessary Steel Phase Diagram Calculations (continued on p. 6 if necessary): 302 Chapter 10/Phase Tranformations in Metals t 10.02. Tural alor diagram for ally steel (ype 4340) A. austen, lie P. pearlie: Mart be (Adapted from I Boyer, Edis, Atherof Fathermal Tr and Cooling Tranformation Di Annican Soci ty for Metals, 1977. p 181) d+y 19.14 0,40whic r 730°C di Fez C 6.77 707°C O aradway is 707°C instead of 727°C/ 800 700 600 500 400- 300 200 100- of 1 Electd temperature Mistart M (50%) 10 ·Pesc @6.69 wt.20 C 10⁰ 10 Time (s) 10 10⁰ 1400 1200 1000 800 600 400 200 10⁰ Temperature (°F) Wt.7. C The presence of alloying elements other than carbon (e.g. Cr. Ni, Mo, and W) may cause significant changes in the positions and shapes of the curves in the isothermal transformation diagrams. These include (1) shifting to longer times the nose of the austenite-to-pearlite transformation (and also a proeutectoid phase nose, it For the phase of diagram-related may be observed by comparing Fig of a separate bainite nose. These alterations calculations the art of proeutectoid product, approximate 4340 steel as being 1040 steel except that the evtec toid temp Figures 10.13 10.14, which are isothermal transformation diagrams for carbon and alloy steels, respectively Steels in which carbon is the prime alloying element are termed plain carbon steels, whereas alloy steels contain appreciable concentrations of other elements, including those cited in the preceding paragraph. Chapter 12 tells more about the classification and properties of ferrous alloys almest the same as EXAMPLE PROBLEM 10.1 Using the isothermal transformation diagram for an iron-carbon alloy of eutee toid composition (Figure 10.13), specify the nature of the final microstructure (in terms of microconstituents present and approximate percentages) of a small specimen that has been subjected to the following time-temperature treatments In each case assume that the specimen begins at 760°C (1400°F) and that it has been held at this temperature long enough to have achieved a complete and homogeneous austenitic structure Temperature (°C) 1700 1500 1300 1100 900 C- 700 500 14 $ 0 4.5% (T) Cu 20 40 Composition (wt% Cu) L TICU Tu 60
Expert Answer:
Answer rating: 100% (QA)
Phase without alloying elements will be Austenite a M rapid cooling b M rapid co... View the full answer

Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-1118324578
9th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these physics questions
-
For a 4340 steel alloy, describe continuous-cooling/tempering heat treatments that would be required to yield specimens having the following yield/tensile strength-ductility property combinations:...
-
A specimen of a 4340 steel alloy having a plane strain fracture toughness of 45 MPa m (41 ksi in.) is exposed to a stress of 1000 MPa (145,000 psi). Will this specimen experience fracture if it is...
-
A specimen of a 4340 steel alloy with a plane strain fracture toughness of 54.8 MPa (50 ksi) is exposed to a stress of 1030 MPa (150,000 psi). Will this specimen experience fracture if the largest...
-
Suppose a town concludes that it costs on average $30.00 per household to manage the disposal of the waste generated by households each year. It is debating two strategies for funding this cost: (1)...
-
A population is modeled by the differential equation dP/dt = 1.2P(1 - P/4200) (a) For what values of P is the population increasing? (b) For what values of P is the population decreasing?
-
Two investments involving a virtual mold apparatus for producing dental crowns qualify for different property classes. Investment A has a cost of \($58,500,\) lasts 9 years with no salvage value, and...
-
Palanquin Auto is a national pre-owned car retailer that offers an online marketplace for customers interested in purchasing a pre-owned automobile. In an effort to reduce price negotiation,...
-
Amoruso Parts Company sells vehicle parts to automotive companies. The Truck Division is organized as a cost center. The budget for the Truck Division for the month ended October 31, 2010, is as...
-
What are the challenges associated with conducting hazard analysis in multi-disciplinary environments, where processes involve a mix of mechanical, electrical, and chemical systems? How can...
-
Consider the following time series data. Quarter Year 1 Year 2 Year 3 1 4 6 7 2 2 3 6 3 3 5 6 4 5 7 8 (a) Choose the correct time series plot. (i) (ii) (iii) (iv) Plot (iv)- Select your answer -Plot...
-
Diego Company manufactures one product that is sold for $80 per unit in two geographic regionsthe East and West regions. The following information pertains to the companys first year of operations in...
-
What is the difference between maintenance work carried out to fix bugs and work carried out to add requested enhancements to a system?
-
Name three mechanisms for creating reusable components in different programming languages.
-
What is the difference between an artefact and a component?
-
Explain why appointments are necessary for many professional services. (Hint: How do services differ from goods as described in Chapter 1?) List and explain some key issues and decisions that must be...
-
Discuss scheduling and sequencing issues in municipal services such as garbage collection, school-bus routing, or snowplowing. What types of criteria and approaches might be used?
-
Determine Current Global Footprint. Present your project company's current global operations. Describe 2-3 Countries that pass Better-Off Test Demand or Supply-Side Scope Economies. Describe the...
-
Tanaka Company's cost and production data for two recent months included the following: March April Production (units).........300................600 Rent.....................$1,800............$1,800...
-
Figure 3.26 shows the first five peaks of the x-ray diffraction pattern for tungsten (W), which has a BCC crystal structure; monochromatic x-radiation having a wavelength of 0.1542 nm was used. (a)...
-
Suppose that a steel of eutectoid composition is cooled to 675C (1250F) from 760C (1400F) in less than 0.5 s and held at this temperature. (a) How long will it take for the austenite-to-pearlite...
-
Give the approximate temperature at which it is desirable to heat each of the following iron-carbon alloys during a full anneal heat treatment: (a) 0.20 wt% C (b) 0.60 wt% C (c) 0.76 wt% C (d) 0.95...
-
What is the direction of the magnetic field at a point vertically (a) above (b) below segment 1 in Figure 28.5? Figure 28.5 Mapping the magnetic field of a current loop. The magnetic field...
-
Make a sketch showing the directions of the magnetic forces exerted on each other by (a) an electron moving in the same direction as the current through a wire, (b) a moving charged particle and a...
-
As the current loop in Figure 28.10 rotates over the first \(90^{\circ}\), do the magnitudes of (a) the magnetic force exerted on the horizontal sides and (b) the torque caused by these forces...

Study smarter with the SolutionInn App


