Aldehydes and Ketones 9. 94 C. Solubility, Iodoform Test, and Benedict's Test 5. Oxidation? (yes/no) 1....
Fantastic news! We've Found the answer you've been seeking!
Question:
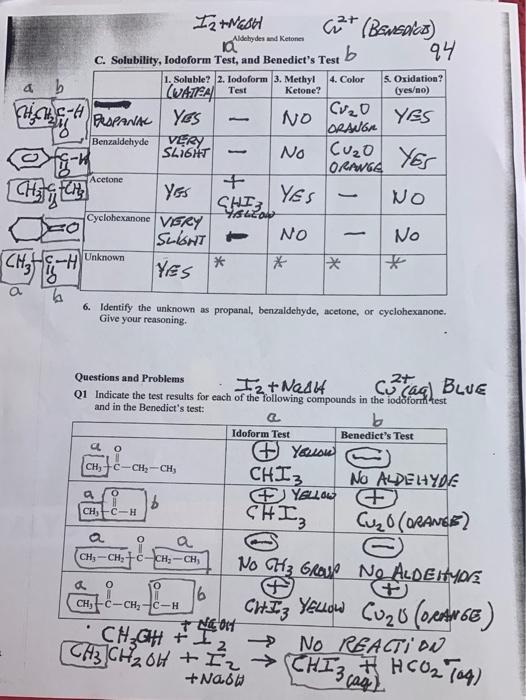
Transcribed Image Text:
Aldehydes and Ketones 9. 94 C. Solubility, Iodoform Test, and Benedict's Test 5. Oxidation? (yes/no) 1. Soluble? 2. lodoform 3. Methyl 4. Color (UATEA Test Ketone? RAPANA YeS Benzaldehyde VERY NO ORANGA YES SLI6HT Cuzo YEr No ORANGE Acctone YES SHI3 NO Cyclobexanone VERY SulGHT NO No Unknown YES 6. Identify the unknown as propanal, benzaldehyde, acetone, or cyclohexanone. Give your reasoning. Questions and Problems ag BLUE Q1 Indicate the test results for each of the following compounds in the iodoform test and in the Benedict's test: Idoform Test Benedict's Test + Yeaou CHI3 (1) No ALDELHYDE CH+C-CH,-CH, O. CH H Cuz0(aRANSE) a a CH-CH;+C-CH;-CH No CH3 GRO No ALDEIMOE 6. CHI Yeuow CH,-C-CH, c-H CHGH t CH3CH2 6H +II→ CHI3, HCO2 Tag) - No REACTI DN Aldehydes and Ketones 9. 94 C. Solubility, Iodoform Test, and Benedict's Test 5. Oxidation? (yes/no) 1. Soluble? 2. lodoform 3. Methyl 4. Color (UATEA Test Ketone? RAPANA YeS Benzaldehyde VERY NO ORANGA YES SLI6HT Cuzo YEr No ORANGE Acctone YES SHI3 NO Cyclobexanone VERY SulGHT NO No Unknown YES 6. Identify the unknown as propanal, benzaldehyde, acetone, or cyclohexanone. Give your reasoning. Questions and Problems ag BLUE Q1 Indicate the test results for each of the following compounds in the iodoform test and in the Benedict's test: Idoform Test Benedict's Test + Yeaou CHI3 (1) No ALDELHYDE CH+C-CH,-CH, O. CH H Cuz0(aRANSE) a a CH-CH;+C-CH;-CH No CH3 GRO No ALDEIMOE 6. CHI Yeuow CH,-C-CH, c-H CHGH t CH3CH2 6H +II→ CHI3, HCO2 Tag) - No REACTI DN
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Give the systematic name for each of the following compounds a. b. (CH3)3CCH2CH2CH2CH(CH3)2 C. d. (CH3CH2)4C e. CH2CH2CH2CH2CH2NHCH2CH3 f. g. h. i. j. k. CH3OCH2CH2CH2OCH3 Br CH3HCH2CH2CHCH2CH2CH3 CH...
-
For each of the following compounds determine which (if any) lone pairs are participating in aromaticity. a. b. c. d. e. f. g. h. :N-H
-
For each of the following compounds determine whether it exhibits a cis configuration or a trans configuration or whether it is simply not stereo-isomeric. a. b. c. d. e. f. g. F
-
The following are the Ledger Balance (in thousands) extracted from the books of Vaishnavi Bank Ltd as on March 31, 2016. The bank's Profit and Loss Account for the year ended and Balance Sheet as at...
-
In a city like New York, the market for stand-up comedians is likely to be monopolistically competitive. Explain why this is. If the market is monopolistically competitive, then what can be said...
-
ERP Projects are expensive and risky. Why did Keda decide to embark on an ERP Implementation Project? What were the main challenges faced by Keda in each stage of its ERP project? What factors...
-
Which certification is most popular among tax accountants? a. Certified Financial Planner b. Certified Public Accountant c. Attorney d. Certified Estate Planner
-
Leased Assets Koffman and Sons signed a four-year lease for a forklift on January 1, 2010. Annual lease payments of $1,510, based on an interest rate of 8%, are to be made every December 31,...
-
Describe examples of business financing that would support short-term assets. What kind of financing would be appropriate for supporting long-term assets? Please explain your answers.
-
Shown as follows is selected information from the financial statements of Dickson, Inc., a retail furniture store. Instructions a. Explain how the interest expense shown in the income statement could...
-
The credit manager of a substantial wholesale trading firm received a visitor who gave his name as Crispin dela Cruz. He was a Sales Engineer in a construction firm who wanted to go business for the...
-
Depending on the nature of R, the reaction of an alkyl bromide, RBr, with cyanide anion, CN, to yield RCN and Br, may proceed via either an SN1 or an SN2 mechanism. What do the terms SN1 and SN2...
-
E-Commerce and its expanded adoption due to the Coronavirus pandemic 1. Name 2 E-commerce companies who benefited and grew during this pandemic? Use these companies and provide valid references...
-
( a ) Describe Holt s method for time series forecast. Write the relevant equations for forecasts. ( a ) Describe Winter s method for time series forecast. Write the relevant equations for the...
-
What is e-commerce and e-business? How an e-commerce business works; Explain the flow chart provided? Identify examples and describe the major types of e-commerce in the mainstream of e-commerce. ...
-
Calculate the NPV for a RIG project that is expected to generate revenues of $1,400,000 per year before tax, in each of the next 10 years. The PV (CCA tax shield) method for depreciation is...
-
Question 23 (Mandatory) (2 points) Larger population tends to shift the labor supply curve rightward at any given wage rate. True False Question 24 (Mandatory) (2 points) Barriers to entry include...
-
In Exercises, find the equation of the tangent line at the given point on each curve. 2y 2 - x = 4; (16, 2)
-
4(3x 5) > 5(2x 3) Describe the solution set as an inequality, in interval notation, and on a graph.
-
4 / 7 b = 5/21
-
1/4b3 1/2 = 9/4
-
If a competitive firm's cost increases due to an increase in the price of a factor of production or a tax, the firm's manager can quickly determine by how much to adjust output by calculating how the...
-
The Canadian metal chair manufacturing market has \(n=78\) firms. The estimated 8.1 elasticity of supply is \(\eta_{0}=3.1\), and the estimated elasticity of demand is \(=-1.1\). Assuming that the...
-
If the cost function for John's Shoe Repair is \(C(q)=100+10 q-q^{2}+\frac{1}{3} q^{3}\), and its marginal cost function is \(M C=10-2 q+q^{2}\), what is its profit-maximizing condition given that...

Study smarter with the SolutionInn App


