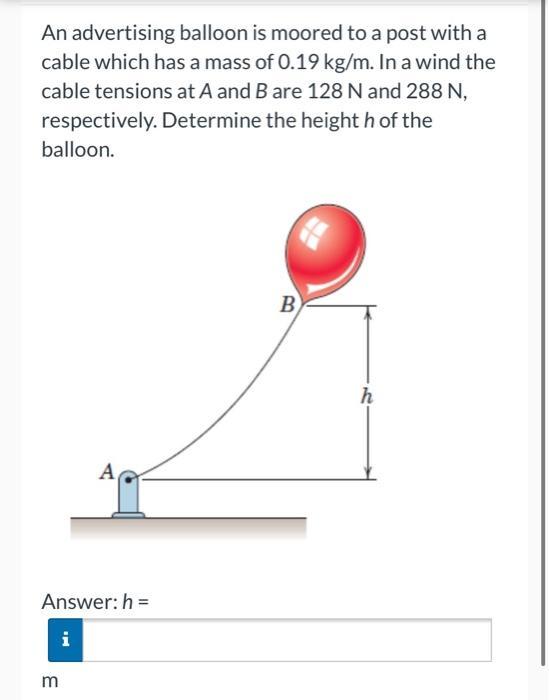
An advertising balloon is moored to a post with a cable which has a mass of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
An advertising balloon is moored to a post with a cable which has a mass of 0.19 kg/m. In a wind the cable tensions at A and B are 128 N and 288 N, respectively. Determine the height h of the balloon. Answer: h= m A IM B h An advertising balloon is moored to a post with a cable which has a mass of 0.19 kg/m. In a wind the cable tensions at A and B are 128 N and 288 N, respectively. Determine the height h of the balloon. Answer: h= m A IM B h
Expert Answer:
Answer rating: 100% (QA)
Solution We have given Mass of the cable per unit length ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The Fashion Rack has a monthly accounting period. The firms chart of accounts is shown below and on the next page. The journals used to record transactions are the sales journal, purchases journal,...
-
A cable with a restrainer at the bottom hangs vertically from its upper end (see figure). The cable has an effective cross-sectional area A = 40 mm2 and an effective modulus of elasticity E = 130...
-
Solve Loe 4 (a) x (b) fo e-y/2 dy 8 X 1+x dx -6 1 ( c ) = = 14 dx (c) - 3 x -
-
Dick Holliday is not sure what he should do. He can build either a large video rental section or a small one in his drugstore. He can also gather additional information or simply do nothing. If he...
-
New Millennium Company earned $2.5 million in net income last year. It took depreciation deductions of $300,000 and made new investments in working capital and fixed assets of $100,000 and $350,000,...
-
Your business uses the allowance method to account for uncollectible receivables. At the beginning of the year, Allowance for Uncollectible Accounts had a credit balance of \($1,100.\) During the...
-
1. John Hood claims that he has no power or authority in his job. Is he correct? What sources of power work for and against him during this change process? 2. What influence tactics has Hood used...
-
A new ALP Government decides to implement more meaningful climate policy. It passes the Safe and Clean Environment Act 2023 (Cth). The legislation regulates a diverse range of activities including...
-
Sharpe Cutter is a small company that produces specialty knives for paper cutting machinery. The annual demand for a particular type of knife is 100,000 units. The demand is uniform over the 250...
-
Other people make decisions way too quickly without considering all the consequences. These people are more impulsive. How you primarily react to the world will affect your decision-making process;...
-
Opticians use the following thin-lens formula to perform calculations related to prescription lenses. = Use this formula to determine i when f= 18 cm and o= 24 cm. Round to the nearest cm.
-
An estate distributes an asset to its sole income beneficiary. The distribution wan, not a specific bequest. The basis of the asset to the estate is $1,000, and the asset's fair market value is...
-
DDD Ltd. is a private corporation with a taxation year ending on December 31. The sole shareholder is Donald Darwin Dorsey who is a Canadian resident throughout 2023. In 2023, DDD Ltd. had the...
-
Write the necessary python code to read the dataset file given to you and perform the following tasks: 1- Detect and list missing data in the dataset. Then fill these missing values with the suitable...
-
The process of obtaining funds from private parties such as investors, venture capital investors, or other companies interested in investing in a business is known as Question 3Answer a. loan...
-
The main issues and challenges that influence the extent to which organisations can manage employment relations to secure high commitment from workers.
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
Phosphorous acid, H3PO3, has the structure (HO)2PHO, in which one H atom is bonded to the P atom, and two H atoms are bonded to O atoms. For each bond to an H atom, decide whether it is polar or...
-
Identify each of the following as an acid or a base in terms of the Arrhenius concept. Give the chemical equation for the reaction of the substance with water, showing the origin of the acidity or...
-
Hydrogen peroxide, H2O2, is a colorless liquid. A concentrated solution of it is used as a source of oxygen for rocket propellant fuels. Dilute aqueous solutions are used as a bleach. Analysis of a...
-
What is the general reporting format on financial statements prepared in accordance with a comprehensive basis of accounting other than generally accepted accounting principles?
-
What is negative assurance within the context of an auditor's report? How does it differ from positive assurance?
-
What body establishes standards for a. All audits and for reviews of interim financial information and preparation of unaudited financial statements of public companies? b. Reviews and compilations...

Study smarter with the SolutionInn App


