An aerosol spray can with a volume of 250 mL contains 2.30 g of propane gas...
Fantastic news! We've Found the answer you've been seeking!
Question:
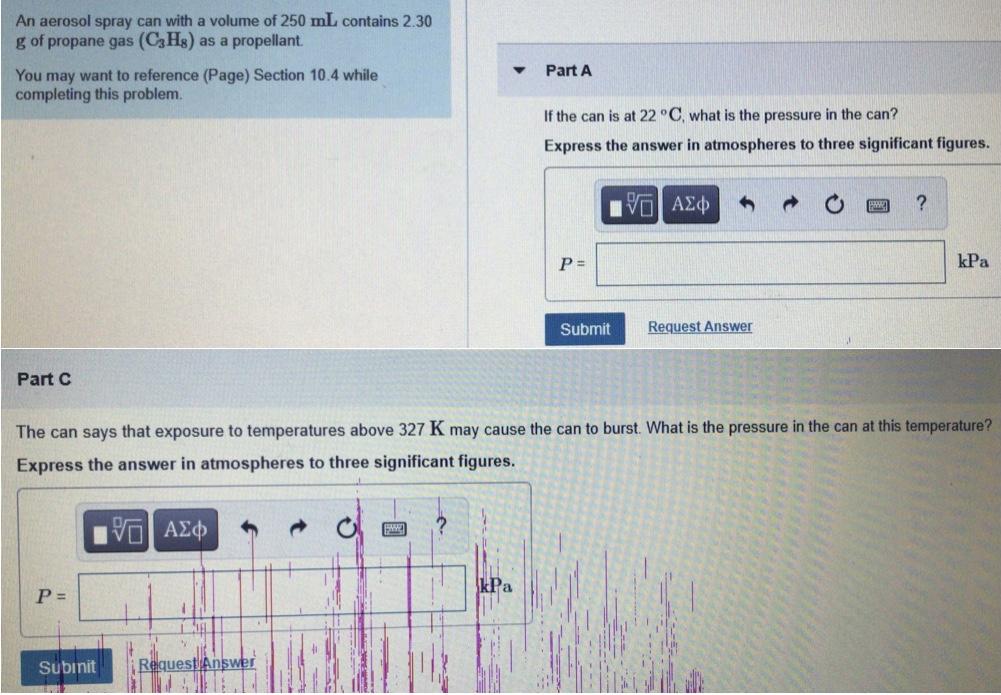
Transcribed Image Text:
An aerosol spray can with a volume of 250 mL contains 2.30 g of propane gas (C3Hs) as a propellant. You may want to reference (Page) Section 10.4 while completing this problem. Part A If the can is at 22 °C, what is the pressure in the can? Express the answer in atmospheres to three significant figures. kPa Submit Request Answer Part C The can says that exposure to temperatures above 327 K may cause the can to burst. What is the pressure in the can at this temperature? Express the answer in atmospheres to three significa igures kPa P = Subinit Request Answer An aerosol spray can with a volume of 250 mL contains 2.30 g of propane gas (C3Hs) as a propellant. You may want to reference (Page) Section 10.4 while completing this problem. Part A If the can is at 22 °C, what is the pressure in the can? Express the answer in atmospheres to three significant figures. kPa Submit Request Answer Part C The can says that exposure to temperatures above 327 K may cause the can to burst. What is the pressure in the can at this temperature? Express the answer in atmospheres to three significa igures kPa P = Subinit Request Answer
Expert Answer:
Answer rating: 100% (QA)
A V 250 mL 025L mass of propane m 230g Molar mass of propane 44gmol n 230 g... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
An aerosol spray can with a volume of 250 mL contain 2.30 g of propane gas (C3H8) as a propellant. (a) If the can is at 23oC, what is the pressure in the can? (b) What volume would the propane occupy...
-
while conducting your analysis you may want to consider and identify the various design elements that are present in the excerpt (ie, use of alignment, chunking, use of color, etc) how effective are...
-
A rigid tank with a volume of 0.117 m3 contains 1 kg of refrigerant-134a vapor at 240 kPa. The refrigerant is now allowed to cool. Determine the pressure when the refrigerant first starts condensing....
-
Calculate the following limits at infinity. If necessary, use -inf and inf to denote -co and co. Use DNE if a limit does not exist. 1. lim e = 818 2. lim e = 8178 3. lim In(x) 24x In(x) =
-
Students in the new MBA class at a state university has the following specialization profile: Finance 67 Marketing 45 Operations and Supply Chain Management 51 Information Systems 18 Find the...
-
Hardness. In a study by J. Zeng et al., three materials for making artificial teethEndura, Duradent, and Duracrosswere tested for hardness. Their results were published as the paper In Vitro Wear...
-
In February 2007, The Elliot Group, Inc., an Illinois real estate developer, made a deal with the Village of Arlington Heights to develop property in that village. Arlington Market, LLC, was...
-
Gilliland Motor, Inc., paid a $3.75 dividend last year. If Gillilands return on equity is 24 percent, and its retention rate is 25 percent, what is the value of the common stock if the investors...
-
With what group of others do you most closely identify? Those from your home town? Those of your same sex? Religion? Race? Income level? IQ level? Who are the people with whom you feel most relaxed?...
-
Applied overhead to Job 201 and to Job 202 using a predetermined overhead rate is 80% of direct materials cost. Transferred Job 201 to Finished Goods Inventory. (1) Sold Job 201 for $167,060 on...
-
The current share price of Winetowat Inc is $50. Future movements in Winetowat Inc's share price are well-described by a continuous process with no jumps and constant volatility. The volatility of...
-
Birdwatcher Inc. manufactures shirts for animal lovers. Its three shirt lines are: Crazy Cat Lady Shirts, Crazy Bird Lady Shirts, and Crazy Squirrel Lady Shirts. The information for these products...
-
Tulip Company presents the following data regarding one of its mixed costs: Month Units Produced Total Cost January 20,000 $30,000 February 40,000 48,000 March 30,000 42,000 April 50,000 63,000 May...
-
In your opinion, what is/are the most critical factors in Controlling Costs in a Project? Use PMBOK to justify your answer.
-
On January 3, 2023, X Corporation borrows $50,000 to buy 10% of the stock in Johnson, Inc. Twenty days later, Johnson, Inc. pays a $1,000 dividend to X Corporation. How much dividend received...
-
Write a complete job description and create compensable factors with job evaluation points for Benefits Manager.
-
You plan to accumulate $448,000 over a period of 25 years by making equal annual deposits in an account that pays an annual interest rate of 12% (assume all payments will occur at the end of each...
-
Which internal control principle is especially diffi cult for small organizations to implement? Why?
-
(a) In which of the following reactions would you expect the orientation factor to be least important in leading to reaction: NO + O NO2 or H + CI HCI? (b) How does the kinetic-molecular theory...
-
(a) The reaction is first H2O2(aq) H2O(I) + O2(g) is first order. Near room temperature, the rate constant equals 7.0 10-4s-1. Calculate the half-life at this temperature. (b) At 415 oC, (CH2)2O...
-
An element X forms an iodide (Xl3) and a chloride (XCl3). The iodide is quantitatively converted to the chloride when it is heated in a stream of chlorine: 2 XI3 + 3 Cl2 2 XCl3 + 3 I2 If 0.5000 g of...
-
Compare and contrast the terms public interest and public benefit in the context of public financial management.
-
Discuss M1, M2 and M3 monetary aggregates.
-
Compare and contrast direct and indirect taxes.

Study smarter with the SolutionInn App


