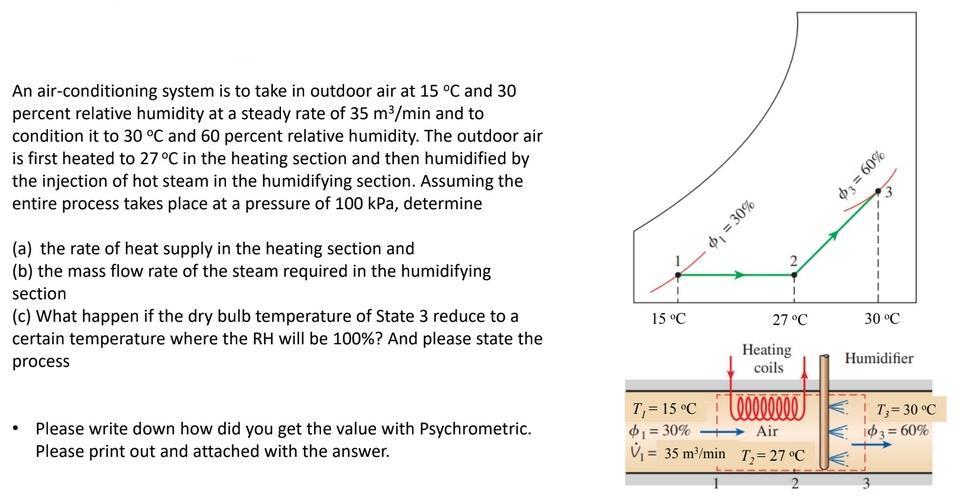
An air-conditioning system is to take in outdoor air at 15 C and 30 percent relative...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
An air-conditioning system is to take in outdoor air at 15 °C and 30 percent relative humidity at a steady rate of 35 m³/min and to condition it to 30 °C and 60 percent relative humidity. The outdoor air is first heated to 27 °C in the heating section and then humidified by the injection of hot steam in the humidifying section. Assuming the entire process takes place at a pressure of 100 kPa, determine (a) the rate of heat supply in the heating section and (b) the mass flow rate of the steam required in the humidifying section (c) What happen if the dry bulb temperature of State 3 reduce to a certain temperature where the RH will be 100% ? And please state the process . Please write down how did you get the value with Psychrometric. Please print out and attached with the answer. 15 °C T₁= 15 °C $₁ = 30% v₁ = $₁ = 30% = 35 m³/min N 27 °C Heating coils teeeeeeee Air T₂= 27 °C 2 %09 = Ep 30 °C Humidifier T₂ = 30 °C 103 = 60% 3 An air-conditioning system is to take in outdoor air at 15 °C and 30 percent relative humidity at a steady rate of 35 m³/min and to condition it to 30 °C and 60 percent relative humidity. The outdoor air is first heated to 27 °C in the heating section and then humidified by the injection of hot steam in the humidifying section. Assuming the entire process takes place at a pressure of 100 kPa, determine (a) the rate of heat supply in the heating section and (b) the mass flow rate of the steam required in the humidifying section (c) What happen if the dry bulb temperature of State 3 reduce to a certain temperature where the RH will be 100% ? And please state the process . Please write down how did you get the value with Psychrometric. Please print out and attached with the answer. 15 °C T₁= 15 °C $₁ = 30% v₁ = $₁ = 30% = 35 m³/min N 27 °C Heating coils teeeeeeee Air T₂= 27 °C 2 %09 = Ep 30 °C Humidifier T₂ = 30 °C 103 = 60% 3
Expert Answer:
Answer rating: 100% (QA)
082 4 307 State 1 T 15C Q 30 h 2307 KJ 3 3 0 082 m 149 Q35m min m 35x1 60 So Enthalpy DBT h 2 ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these physics questions
-
An air conditioning system is to take in air at 1 atm, 32oC, 65% relative humidity and deliver it at 22oC, 40% relative humidity. Air flows first over the cooling coils, where it is cooled and...
-
An air-conditioning system is to take in air at 1 atm, 34C, and 70 percent relative humidity and deliver it at 22C and 50 percent relative humidity. The air flows first over the cooling coils, where...
-
An air conditioning system operates at a total pressure of 1 atm consists of a heating section and humidifier which supplies wet steam (saturated water vapor) at 1 atm. Air enters the heating section...
-
Paul made the following acquisitions and disposals of ordinary shares in Crimmon plc: No further shares were acquired during 2020. Against which acquisitions will each of the above disposals be...
-
A 30-mm-diameter copper rod is 1 m long with a yield strength of 70 MPa. Determine the axial force necessary to cause the diameter of the rod to reduce by 0.01 percent, assuming elastic deformation....
-
Clarissa's Cupcakes provides you with the following data for March: ............................................Master Budget............... Actual Result Sales (# of...
-
Locate sample affidavits used in a civil litigation for both California court and federal court. What do they have in common? Are there any striking differences?
-
Michael and Nora Hoth married, divorced, remarried, divorced again, then cohabited from 1998 to 2008 without further remarriage. In 2006, Michael bought a house and five acres with his own money....
-
Suppose a stock had a value of $28.23 per share at the end of 2007. And suppose the stock paid $2 in dividends at the end of 2008 and $2.25 at the end of 2009. If the stock's value is $30 at the end...
-
Consider a specimen of ice that is at 210C and 1 atm pressure. Using Figure 9.2, the pressure-temperature phase diagram for H2O, determine the pressure to which the specimen must be raised or lowered...
-
Let S(0) 100, R = 0.05, U = 0.2, D = -0.2, and N = 3, and consider the payoff of following European butterfly spread: -30- -20- -10- 50 = 60 70 80 90 100 110 120 130 140 (a) You can construct this...
-
Abbott Company completed its bank reconciliation and needs to record a $30 service charge from its bank. What would be the journal entry to record this transaction?
-
Solve each problem. Use the information from the graph to figure out how much the government pays in price supports to this farmer for soybeans. Soybean Supply/Demand Curve Price ($) 7.00 Dernand...
-
# INSERT YOUR CODE HERE # # 5. Write your code to construct a box plot for the COVID mortality total_deaths_per_million. #(3 marks) #Refer to Week 4 practical for plotting graphs.
-
In a particle accelerator, a magnetic force keeps a lithium nucleus (mass 6.02 u) traveling in a circular path with a radius of 0.530 m. The lithium nucleus moves at a speed of 10.50% of the speed of...
-
Image caption a) Set the Q-point of each transistor to be in the middle of the forward active region. b) Calculate the DC voltage at the input and output points of each amplifier. AC ANALYSIS OF THE...
-
Create a program called PIK-PAK-BOOM. The program prints the numbers from the specified start number to the end number, printing n numbers per line. It shall print "Pik" in place of the numbers which...
-
Banner Company acquires an 80% interest in Roller Company for $640,000 cash on January 1, 2013. The NCI has a fair value of $160,000. Any excess of cost over book value is attributed to goodwill. To...
-
A gas-turbine power plant operates on the regenerative Brayton cycle between the pressure limits of 100 and 700 kPa. Air enters the compressor at 308C at a rate of 12.6 kg/s and leaves at 2608C. It...
-
A piston - cylinder device contains steam initially at 1 MPa, 450oC, and 2.5 m3. Steam is allowed to cool at constant pressure until it first starts condensing. Show the process on a T v diagram with...
-
Steam enters an adiabatic turbine at 6 MPa, 600C, and 80 m/s and leaves at 50 kPa, 100C, and 140 m/s. If the power output of the turbine is 5 MW, determine (a) The reversible power output and (b) The...
-
A stirrer-container assembly contains a certain amount of fluid. The stirrer performs \(3 \mathrm{hp}\) work on the system. The heat developed by stirring is \(4000 \mathrm{~kJ} / \mathrm{h}\) and is...
-
Derive the mathematical expression of the first law of thermodynamics.
-
A system consisting of a gas confined in a cylinder undergoes a series of processes shown in Fig. 2.11. During the process A-1-B, \(70 \mathrm{~kJ}\) of heat is added while it does 45 \(\mathrm{kJ}\)...

Study smarter with the SolutionInn App


