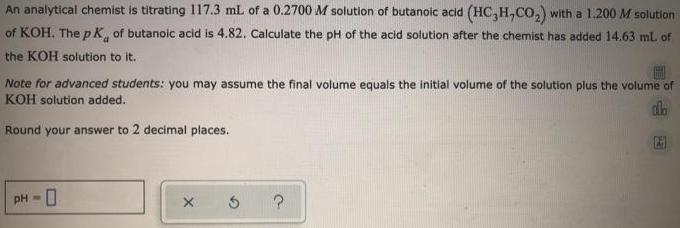
An analytical chemist is titrating 117.3 ml of a 0.2700 M solution of butanoic acid (HC,H,CO,)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
An analytical chemist is titrating 117.3 ml of a 0.2700 M solution of butanoic acid (HC,H,CO,) with a 1.200 M solution of KOH, ThepK, of butanoic acid is 4.82. Calculate the pH of the acid solution after the chemist has added 14.63 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added. Round your answer to 2 decimal places. pH = An analytical chemist is titrating 117.3 ml of a 0.2700 M solution of butanoic acid (HC,H,CO,) with a 1.200 M solution of KOH, ThepK, of butanoic acid is 4.82. Calculate the pH of the acid solution after the chemist has added 14.63 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added. Round your answer to 2 decimal places. pH =
Expert Answer:
Answer rating: 100% (QA)
Concentration of butanoic acid HC3H7CO2 02700M Volume of butanoic acid taken 1173 mL Concentration o... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Complete the following ANOVA table. *Round your answer to 2 decimal places, the tolerance is +/-0.01. **Round your answer to 3 decimal places, the tolerance is +/-0.001. *** Round your answer to 2...
-
An analytical chemist is titrating 53.9 mL of a 0.6000 M solution of trimethylamine ((CH,),N) with a 0.7900 M solution of HNO3. The p K, of 3. trimethylamine is 4.19. Calculate the pH of the base...
-
An analytical chemist is titrating 60.6 mL of a 0.5600 M solution of formic acid (H,CO,) with a 0.3100 M solution of KOH. The p K, of formic acid is 3.74. Calculate the pH of the acid solution after...
-
What are the Marketing Cost Estimates of Pepsi Company? Marketing estimates, in 2013-2019? It can be write in a paragraph and explain it statistically.
-
What assumptions underlie bivariate regression?
-
The shoe sizes (such as 8 or 101 2) of test subjects. Determine whether the data described are qualitative or quantitative and explain why.
-
Following is a performance report for Electric Hero. Required (a) Calculate the variances, stating whether they are favourable or unfavourable, and suggest possible reasons for variances that you...
-
During 2012 and 2013, Gupta Co. completed the following transactions relating to its bond issue. The companys fiscal year ends on December 31. 2012 Mar. 1 Issued $100,000 of eight-year, 7 percent...
-
Below are estimates related to Sando's 2022 budget, a company that specializes in crafting unique ornaments for the Caribbean: Selling Price: $2500 Variable Cost per Ornament: $1625 Fixed Annual...
-
There is a parcel of land next to the Playful Paws, Inc. building. Ellen, the owner of this property, approached John to discuss the idea of selling it to him. John is interested. Ellen knows that...
-
what are the interrelationships between the building block of a Sauna Suit business?
-
Why is the stop method for stopping a thread deprecated? How do you terminate a thread?
-
Write the XML document that corresponds to the parse tree in Figure 2. Figure 2 Left shift ( < >) 0 0 Right shift with zero extension (>>>) 0
-
An orthopedic group in Virginia has decided to conduct a survey of referral physicians. The results, which are posted on the group's website, indicate the level of customer satisfaction. The group...
-
Write a program that reads all lines from a file and, using a Stream , prints how many of them contain the word the.
-
What happens if you try to save an object that is not serializable in an object output stream? Try it out and report your results.
-
what decision refers to the selection of the best means to achieve a goal and not to the selection of the goal itself. If an individual wishes to own a car primarily as a means of transportation,...
-
A stock has had returns of 8 percent, 26 percent, 14 percent, 17 percent, 31 percent, and 1 percent over the last six years. What are the arithmetic and geometric average returns for the stock?
-
Ethylene glycol is used as an automobile antifreeze and in the manufacture of polyester fibers. The name glycol stems from the sweet taste of this poisonous compound. Combustion of 6.38 mg of...
-
The root-mean-square speed of the molecules of an ideal gas at 25oC and a pressure of 2.50 atm is 5.00 102 m/s. What is the density of this gas?
-
A solution containing calcium ion and magnesium ion is buffered with ammonia-ammonium chloride. When carbonate ion is added to the solution, calcium carbonate precipitates but magnesium carbonate...
-
Consider the multiple linear regression model fit to the baseball data in Problem 3.41. Problem 3.41 Consider the 2016 major league baseball data in Table B.22. While team ERA was useful in...
-
Consider the simple linear regression model fit to the rental price data from Problem 2.36. Data From Problem 2.36 Table B.24 contains data on median family home rental price and other data for 51 US...
-
Consider the multiple linear regression model fit to the rental price data in Problem 3.42. Problem 3.42 Table B.24 contains data on median family home rental price and other data for 51 US cities....

Study smarter with the SolutionInn App


