Question: An atom in its single state is subjected to a magnetic field. The Zeeman splitting of its 4500 A spectral lines is 14 m.
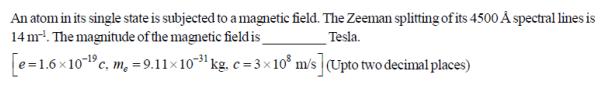
An atom in its single state is subjected to a magnetic field. The Zeeman splitting of its 4500 A spectral lines is 14 m. The magnitude of the magnetic field is Tesla. [e=1.6x10-1c c. m, = 9.1110- kg. c=3108 m/s] (Upto two decimal places)
Step by Step Solution
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The detailed ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


