An organic compound is burnt with excess of O to produce CO(g) and HO(1), which results...
Fantastic news! We've Found the answer you've been seeking!
Question:
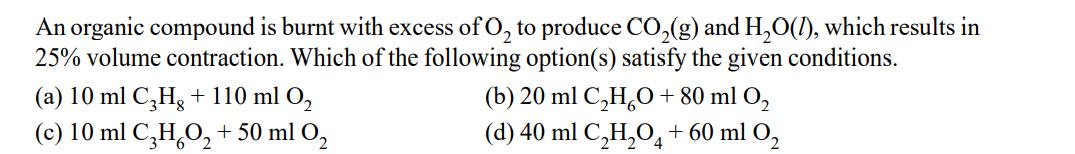
Transcribed Image Text:
An organic compound is burnt with excess of O₂ to produce CO₂(g) and H₂O(1), which results in 25% volume contraction. Which of the following option(s) satisfy the given conditions. (a) 10 ml C3Hg + 110 ml O₂ (c) 10 ml C3H₂O₂ + 50 ml O₂ (b) 20 ml C₂H₂O + 80 ml 0₂ (d) 40 ml C₂H₂O4 + 60 ml O₂ An organic compound is burnt with excess of O₂ to produce CO₂(g) and H₂O(1), which results in 25% volume contraction. Which of the following option(s) satisfy the given conditions. (a) 10 ml C3Hg + 110 ml O₂ (c) 10 ml C3H₂O₂ + 50 ml O₂ (b) 20 ml C₂H₂O + 80 ml 0₂ (d) 40 ml C₂H₂O4 + 60 ml O₂
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A quantity of 7.480 g of an organic compound is dissolved in water to make 300.0 mL of solution. The solution has an osmotic pressure of 1.43 atm at 27C. The analysis of this compound shows that it...
-
An organic compound is analyzed and found to contain 66.7% carbon, 11.2% hydrogen, and 22.1% oxygen by mass. The compound boils at 79.6C. At 100C and 0.970 atm, the vapor has a density2.28 g/L. of....
-
An organic compound is found to contain 37.5 percent carbon, 3.2 percent hydrogen, and 59.3 percent fluorine by mass. The following pressure and volume data were obtained for 1.00 g of this substance...
-
Debate on Causes of Unemployment Two economists are debating the cause of a high unemployment rate. One economist argues that there is not enough government spending. The other says high unemployment...
-
You are the manager in charge of global operations at Bank Globala large commercial bank that operates in a number of countries around the world. You must decide whether or not to launch a new...
-
You are the design engineer in charge of the crashworthiness of new automobile models. Cars are tested by smashing them into fixed, massive barriers at 45 km/h. A new model of mass 1500 kg takes 0.15...
-
On a brilliant, sunny day, you take a long hike through open country. You sweat a lot, losing a lot of water. What hormone does your body release? Why?
-
The figure shows a shaft mounted in bearings at A and D and having pulleys at B and C. The forces shown acting on the pulley surfaces represent the belt tensions. The shaft is to be made of ASTM...
-
You are managing a restaurant. You do not accept reservations, and so all tables are walk-in. Customers that arrive and request a table are divided as follows: 50% require a table for 2, 40% require...
-
1. What are the basic elements to consider in the case of Mergers and Acquisitions? 2. How can you convince your shareholders about a possible Merger? 2. Hilton Hotel is considering, in the annual...
-
Blossom Co. sells product P-14 at a price of $49 a unit. The per-unit cost data are direct materials $15, direct labour $10, and overhead $12 (75% variable). Blossom Co. has sufficient capacity to...
-
A new project being considered by BLW Co would require 1,000 hours of skilled labour. The current workforce is already fully employed but more workers can be hired in at a cost of $20 per hour. The...
-
Sales Returns and Allowances On December 10, Year 1, Cantlay Inc. made $500,000 in sales on account to various customers. The cost of the merchandise sold is $350,000. Cantlay estimates that 4% of...
-
Mr Lee commenced his business on 1 November 2014 and purchased the following asset in the first 17 months of operations: Date of purchase Purchase price /cost 2014 $ 1 November One second-hand motor...
-
The ___ operator reverses the truth of its Boolean operand. and or mod not$150,000 6% Bonds were issued October 1, 2022 for $129,555. Semiannual interest payments are due March 31 and September 30....
-
A 1100-V, 50-Hz, 4 pole delta-connected induction motor has a star-connected slip- ring rotor with a phase turns ratio of 3.8:1. The rotor resistance and standstill leakage reactance are 0.012 2 and...
-
Silver Star's Foreign Markets hedge fund manages an S&P 500 stock index fund worth $118 million. Assume that the S&P 500 index realized a mean monthly return of 3.6% in the period January 2011...
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
Referring to Problem 18.46, explain why the ratio is considerably smaller than 90 J/K ? mol for liquid HF.
-
Predict the pH (7 >, < 7, or 72 of aqueous solutions containing the following salts: (a) KBr, (b) Al(NO3)3, (c) BaCl2, (d) Bi(NO3)3.
-
Calcium oxide or quicklime (CaO) is used in steelmaking, cement manufacture, and pollution control. It is prepared by the thermal decomposition of calcium carbonate: CaCO3(s) CaO(s) + CO2(g)...
-
In testing the null hypothesis that p = 0:3 against the alternative that p 0:3, the probability of a type II error is ______ when the true p = 0:4 than when p = 0:6. (1) the same (2) smaller (3)...
-
In a hypothesis test the p value is 0.043. This means that we can find statistical significance at: (1) both the 0.05 and 0.01 levels (2) the 0.05 but not at the 0.01 level (3) the 0.01 but not at...
-
An article states there is no significant evidence that median income increased. The implied null hypothesis is: (1) Median income increased. (2) Median income changed. (3) Median income did not...

Study smarter with the SolutionInn App


