Question: 11. The differential equation which has y = C1+ C2a3 as its general solution is: (a) ry 2y' = 0 (b) xy 2ry +
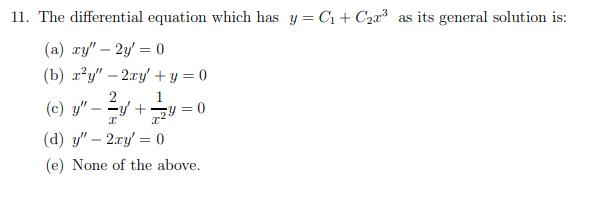
11. The differential equation which has y = C1+ C2a3 as its general solution is: (a) ry" 2y' = 0 (b) xy" 2ry + y = 0 2 1 (c) y" - y + (d) y" 2ry' = 0 (e) None of the above.
Step by Step Solution
3.50 Rating (163 Votes )
There are 3 Steps involved in it
I have solved t... View full answer

Get step-by-step solutions from verified subject matter experts


