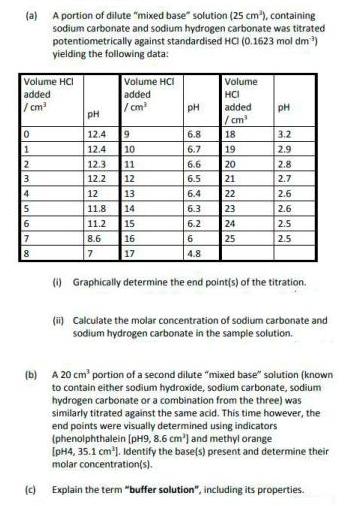
(a) A portion of dilute mixed base solution (25 cm), containing sodium carbonate and sodium hydrogen...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(a) A portion of dilute "mixed base" solution (25 cm), containing sodium carbonate and sodium hydrogen carbonate was titrated potentiometrically against standardised HO (0.1623 mol dm) yielding the following data: Volume HCI added / cm Volume HCI added /cm Volume HCI added / cm 18 19 pH pH pH 12.4 19 6.8 3.2 12.4 10 11 6.7 2.9 12.3 6.6 20 2.8 21 22 23 24 25 3 12.2 12 6.5 2.7 4 12 13 6.4 2.6 11.8 14 6.3 2.6 15 16 17 6 11.2 6.2 2.5 7 8.6 6 2.5 8 7 4.8 (i) Graphically determine the end point(s) of the titration. (i) Calculate the molar concentration of sodium carbonate and sodium hydrogen carbonate in the sample solution. (b) A 20 cm' portion of a second dilute "mixed base" solution (known to contain either sodium hydroxide, sodium carbonate, sodium hydrogen carbonate or a combination from the three) was similarly titrated against the same acid. This time however, the end points were visually determined using indicators (phenolphthalein [pH9, 8.6 cm') and methyl orange [pH4, 35.1 cm). Identify the base(s) present and determine their molar concentration(s). (c) Explain the term "buffer solution", including its properties. (a) A portion of dilute "mixed base" solution (25 cm), containing sodium carbonate and sodium hydrogen carbonate was titrated potentiometrically against standardised HO (0.1623 mol dm) yielding the following data: Volume HCI added / cm Volume HCI added /cm Volume HCI added / cm 18 19 pH pH pH 12.4 19 6.8 3.2 12.4 10 11 6.7 2.9 12.3 6.6 20 2.8 21 22 23 24 25 3 12.2 12 6.5 2.7 4 12 13 6.4 2.6 11.8 14 6.3 2.6 15 16 17 6 11.2 6.2 2.5 7 8.6 6 2.5 8 7 4.8 (i) Graphically determine the end point(s) of the titration. (i) Calculate the molar concentration of sodium carbonate and sodium hydrogen carbonate in the sample solution. (b) A 20 cm' portion of a second dilute "mixed base" solution (known to contain either sodium hydroxide, sodium carbonate, sodium hydrogen carbonate or a combination from the three) was similarly titrated against the same acid. This time however, the end points were visually determined using indicators (phenolphthalein [pH9, 8.6 cm') and methyl orange [pH4, 35.1 cm). Identify the base(s) present and determine their molar concentration(s). (c) Explain the term "buffer solution", including its properties.
Expert Answer:
Answer rating: 100% (QA)
From the graph with xaxis representing volume of acid and Yaxis representing pH values First inflect... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given an acceleration vector, initial velocity (uo,vo), and initial position (Xo.Yo), find the velocity and position vectors for t>0. a(t) = (0,1), (Uo.vo) = (5,7), (Xo.Yo) = (0,0) %3D %3D What is...
-
Calculate the mode, mean, and median of the following data: 18 10 15 13 17 15 12 15 18 16 11
-
Sodium hydrogen carbonate is also known as baking soda. When this compound is heated, it decomposes to sodium carbonate, carbon dioxide, and water vapor. Write the balanced equation for this...
-
4. Using Euler's method to solve following equation with time step of 1. dy = = 4t - 0.5y dt y(0)=2 You only need to write out three steps to get values of y(1), y(2), y(3). 5. Write your Euler's...
-
Patty's Pizza Parlor initially had the production function given in the table in Problem 3. A worker's hourly wage rate was $10, and pizza sold for $2. Now Patty buys a new high-tech pizza oven that...
-
In a neighborhood challenge to see who can climb a tree the fastest, you are ready to climb. Your friends have surrounded you in a circle as a cheering section; each individual alone would cause a...
-
Assume you are considering internal control over a client's inventory and fixed asset records. System documentation was accomplished with a flowchart and questionnaire and, in conjunction with a...
-
During 2011 the following selected transactions affecting stockholders equity occurred for Orlando Corporation: a. Apr. 1 Purchased in the market 200 shares of the companys own common stock at $20...
-
Create a popsicle stick bridge that is 12 inches long and 3inches high, with no width restriction BUT it must be a threedimensional truss/bridge. You can only use 25 popsicle sticks, but they can be...
-
Frank is looking at a new sausage system with an installed cost of $560,000. This cost will be depreciated straight line to zero over the projects five-year life, at the end of which the sausages...
-
3. (a) Prove by the Principle of Mathematical Induction that n? +n is divisible by 2 for any n E N*. (6 marks) (b) Consider the sequence of real numbers In = Prove by definition that rm converges to...
-
The units of an item available for sale during the year were as follows: Jan. 1 Aug. 13 Inventory Purchase Nov. 30 Purchase Available for sale 17 units at $24 $408 8 units at $25 2001 10 units at $26...
-
With an increasing focus on bullying among the public, educators, researchers, and policy makers, there is a need to delineate what we know works in bullying prevention and what areas might benefit...
-
What is the wavelength in meters of a lightwave with a frequency of 1 1 0 khz ?
-
Becker Tabletops has two support departments (Janitorial and Cafeteria) and two production departments (Cutting and Assembly). Relevant details for these departments are as follows: Support...
-
a. Received $7,300 in cash and checks from a door-to-door campaign. b. Incurred and paid $2,200 cash for employee wages this month. c. Paid $3,700 cash on a short-term loan from the bank (ignore...
-
how does 25000 of common stock issued at .98 affect common stock outstanding of 100000?
-
Economic feasibility is an important guideline in designing cost accounting systems. Do you agree? Explain.
-
Boric acid, H3BO3, is a mild antiseptic and is often used as an eyewash. A sample contains 0.543 mol H3BO3. What is the mass of boric acid in the sample?
-
At standard temperature and pressure, a 1.00-mol sample of argon gas is vented into a 22.4-L rigid box that already contains 1.00 mol of nitrogen gas. We would expect the argon gas to a. decrease the...
-
Calculate the following. a. Number of atoms in 25.7 g Al b. Number of atoms in 8.71 g I2 c. Number of molecules in 14.9 g N2O5 d. Number of formula units in 3.31 g NaClO4 e. number of Ca2+ ions in...
-
An electronic instrument is to be isolated from a panel that vibrates at frequencies ranging from \(25 \mathrm{~Hz}\) to \(35 \mathrm{~Hz}\). It is estimated that at least 80 percent vibration...
-
An exhaust fan, having a small unbalance, weights \(800 \mathrm{~N}\) and operates at a speed of \(600 \mathrm{rpm}\). It is desired to limit the response to a transmissibility of 2.5 as the fan...
-
The armature of a variable-speed electric motor, of mass \(200 \mathrm{~kg}\), has an unbalance due to manufacturing errors. The motor is mounted on an isolator having a stiffness of \(10 \mathrm{kN}...

Study smarter with the SolutionInn App


