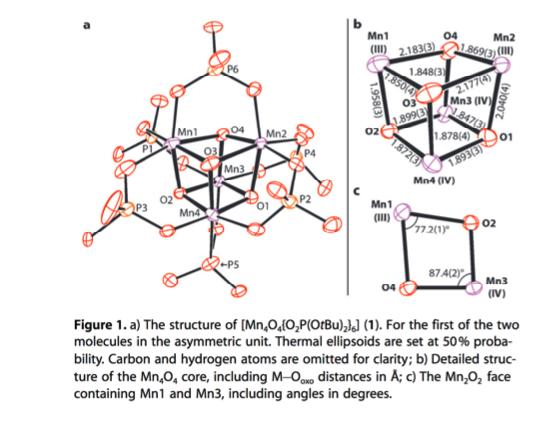
A cluster complex, Mn 4 O 4 (L) 6 (L = [P(O t Bu) 2 O 2
Question:
A cluster complex, Mn4O4(L)6 (L = [P(OtBu)2O2–]), was reported by Van Allsburg, et al. in Chem. Eur. J. 2015, 21, 4646.
What is the average oxidation state of Mn is this complex? Show your work.
The crystal structure of this cluster was solved, and the bond metrics of the [Mn4O4] unit are given below (Figure 1). The authors determined that the oxidation state of each Mn ion is best described with integer values (MnIII (+3) and/or MnIV (+4)), meaning the oxidation states are localized on each metal rather than averaged over the cluster. Explain how the authors concluded from the given data which Mn ions would be labeled as MnIII or MnIV (Figure 1b)?

Applying International Financial Reporting Standards
ISBN: 978-0730302124
3rd edition
Authors: Keith Alfredson, Ken Leo, Ruth Picker, Paul Pacter, Jennie Radford Victoria Wise




