Question: Apply bubble sort to the following list to sort it in ascending order. Record your answer for the first two passes. 29 73 43
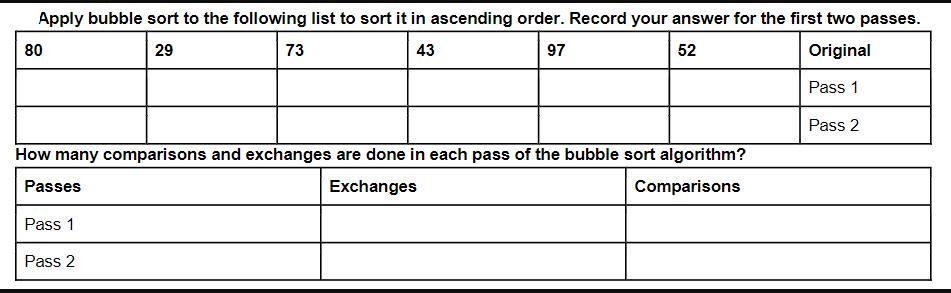
Apply bubble sort to the following list to sort it in ascending order. Record your answer for the first two passes. 29 73 43 97 52 Original Pass 1 80 How many comparisons and exchanges are done in each pass of the bubble sort algorithm? Passes Exchanges Comparisons Pass 1 Pass 2 Pass 2
Step by Step Solution
There are 3 Steps involved in it
1 ans selection sort 80 29 29 passes pass1 pass2 80 29 29 ... View full answer

Get step-by-step solutions from verified subject matter experts


