Arcelor Corp issues $400,000, 6%, 10-year bonds. The market rate at the date of issue is...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
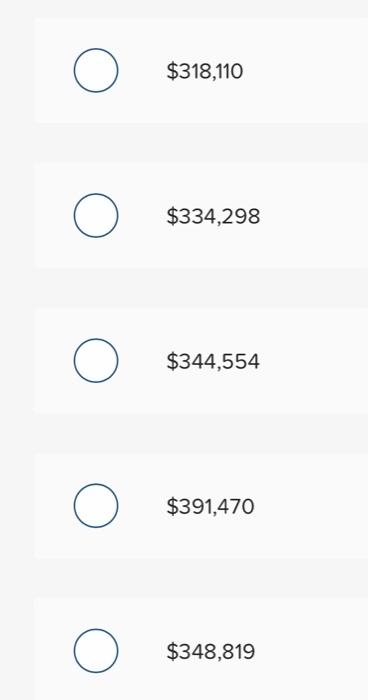
Arcelor Corp issues $400,000, 6%, 10-year bonds. The market rate at the date of issue is 5.5%. The total amount of interest to be paid to the bondholders for the semiannual interest payments, assuming the effective interest method is used, is $318,110 $334,298 $344,554 $391,470 $348,819 Arcelor Corp issues $400,000, 6%, 10-year bonds. The market rate at the date of issue is 5.5%. The total amount of interest to be paid to the bondholders for the semiannual interest payments, assuming the effective interest method is used, is $318,110 $334,298 $344,554 $391,470 $348,819
Expert Answer:
Answer rating: 100% (QA)
X V fr SUMD6D25 D26 A B D F issue price of bonds 415227 2 interest interes... View the full answer

Related Book For 

Governmental and Nonprofit Accounting
ISBN: 978-0132751261
10th edition
Authors: Robert Freeman, Craig Shoulders, Gregory Allison, Robert Smi
Posted Date:
Students also viewed these accounting questions
-
What is the maximum amount of interest to be capitalized under IFRS?
-
The effective interest method assigns a bond interest expense amount that increases over the life of a premium bond
-
Assuming no interest is paid on checking accounts, what would you expect to see happen to the relative growth rates of M1 and M2 if interest rates rose significantly?
-
According to the Mortgage Bankers Association, 10% of all home mortgage loans in California were in foreclosure in the fourth quarter of 2011. Assuming this rate holds, if you randomly pick 30...
-
Shown here are the data for y and three predictors, x1, x2, and x3.A multiple regression analysis has been done on these data; the Minitab results are given. Comment on the outcome of the analysis in...
-
Few brands dominate their industry with a more than 50 percent global market share. Gillette did that for decades, long the razor market leader, with veteran brands Schick and Bic running a distant...
-
The salaries (in thousand dollars) of a sample of 10 employees Find the mean, the median, and the mode of the data, if possible. If any measure cannot be found or does not represent the center of the...
-
The national stock exchanges require listed companies to have an independent audit committee. Required a. Describe an audit committee. b. What are the typical functions performed by an audit...
-
Consider a project that requires an investment of $1700 in the first year, but will generate a NPV of $80. Calculate the profitability index value of this project.
-
The information below pertains to the retiree health care plan of Thompson Technologies: Thompson began funding the plan in 2024 with a contribution of $127,000 to the benefit fund at the end of the...
-
ABC Company plans to discontinue a product line. The product line currently generates sales revenue of $100,000 and total contribution margin of $50,000 annually. The fixed costs associated with the...
-
The matrices in the state-space form of a system model are given. a. Find the transfer function (or transfer matrix) by using Equation 4.21. b. Verify the result of (a) by using the ss2tf command....
-
A flue gas consists of carbon dioxide, oxygen, water vapor, and nitrogen. The molar fractions of \(\mathrm{CO}_{2}\) and \(\mathrm{O}_{2}\) in a sample of the gas are \(12 \%\) and \(6 \%\),...
-
A solution of carbon tetrachloride and carbon disulfide containing \(50 \%\) by weight of each is to be continuously distilled at the rate of \(5000 \mathrm{~kg} / \mathrm{h}\). (a) Determine the...
-
A mixture of noble gases (helium, argon, krypton, and xenon) is at a total pressure of \(150 \mathrm{kPa}\) and a temperature of \(500 \mathrm{~K}\). If the mixture has equal mole fractions of each...
-
Air, stored in a \(30-\mathrm{m}^{3}\) container at \(340 \mathrm{~K}\) and \(150 \mathrm{kPa}\), is saturated with water vapor. Determine the following properties of the gas mixture: (a) Mol...
-
How well employees modify their thoughts and behavior to align with and support a new or changing environment is known as Multiple Choice proactive task performance. proficient task performance....
-
For what reason might an exporter use standard international trade documentation (letter of credit, draft, order bill of lading) on an intrafirm export to its parent or sister subsidiary?
-
Multiple Choice Questions Identify the best answer to each question: 1. Each of the following criteria is a factor when determining Special Revenue Fund property tax revenue recognition except: a....
-
Internal Service Funds are required to be reported by fund type in the basic financial statements. Discuss the significance of this requirement. Compare and contrast the financial reporting required...
-
In what funds and nonfund accounts are (a) Capital assets (b) Long-term liabilities accounted for?
-
Eq. 7.36a is written for the conservation of momentum in y direction. Obtain Eq. 7.36-b wherein the stream function is independent variable. Eq. 7.36(a,b) y - v v +(1-y/R)v- + momentum: u u R-y + R P...
-
Using Maslen method, find the approximate value of pressure and density at the junction of the sphere and the cone of Problem 7.29 at Mach number 8. Problem 7.29 An empirical way to determine shock...
-
Show that the derivative of the boundary layer edge velocity is given by Eq. 7.64 for the figure given below. Eq. 7.64 M>>1 Ue dx R dy/dx = 1/R

Study smarter with the SolutionInn App


