1. (30%) As a part of the liquefaction process ammonia goes through an ideal compressor and...
Fantastic news! We've Found the answer you've been seeking!
Question:

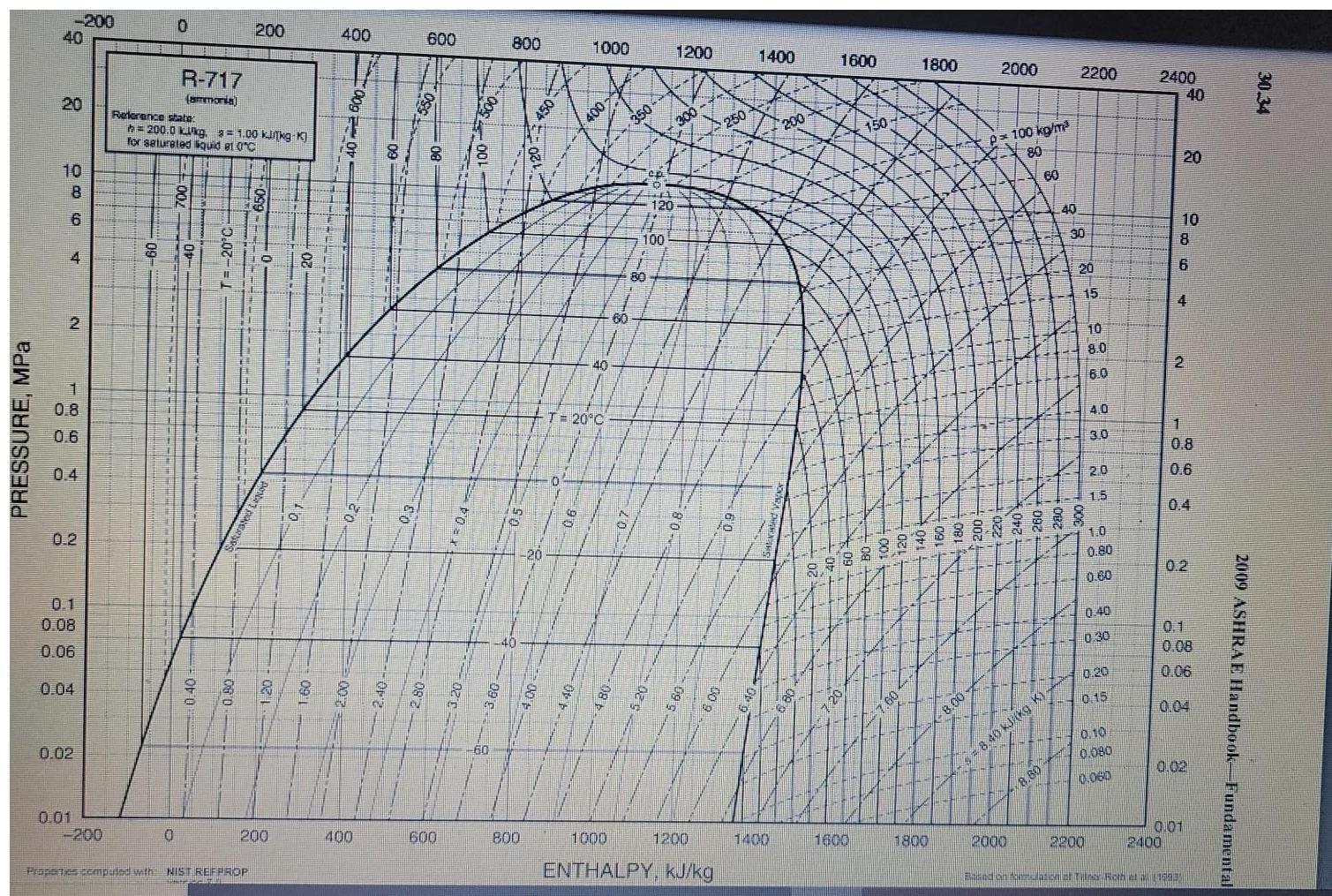
Transcribed Image Text:
1. (30%) As a part of the liquefaction process ammonia goes through an ideal compressor and a heat exchanger. After that supercritical ammonia goes through a throttling valve. Ammonia en ters the compressor at T₁ = 60°C and P₁ = 20 bar and is compressed to the pressure P₁ = 200 bar, and then cooled down in the heat exchanger to T3 = 140°C and flashed at the pressure P = 1 bar Use the attached PH diagram for ammonia to answer the following questions: A. (10) Draw this process on the attached PH diagram. B. (5) Calculate the fraction of ammonia liquefied. C. (5) Calculate the work performed in the compressor. D. (5) Calculate the amount of heat rejected in the heat exchanger. E. (5) How would the fraction of ammonia liquefied change if the amount of heat rejected in the heat exchanger remained the same, but the compressor is replaced with a non-ideal one? (Briefly explain). PRESSURE, MPa -200 40 20 10 8 6 4 2 1864 0.8 0.6 0.2 0.1 0.08 0.06 0.04 0.01 0.02 -200 60 0 0 R-717 (ammonia) Reference state h=200.0 kkg. = 1.00 kinkg-K] for saturated liquid et 0°C 0.40 200 Properties computed with NIST REFPROP O PONA 200 OB 400 20 400 600 8 8 600 500 800 15 6 40 800 450 1000 400 140 T= 20°C 250 60 ep 120 EL 100 1200 300 9 1200 3 100 1000 ENTHALPY, kJ/kg 250 1400 1400 200 1600 1600 150 1800 1800 8.00- 2000 p=100 kg/m³ 80 60 2000 4-5 = 8.40 kJ/(kg K) 40 8.80 2200 30 20 15 10 8.0 6.0 4.0 3.0 2.0 15 1.0 0.80 0.60 2200 0.40 0:30 0.20 0.15 0.10 0.080 0.060 2400 10 8 6 40 20 4 2 2400 1 0.8 06 0.2 0.08 0.06 0.04 0.01 0.02 and on fostulation af Tile Roth (1203 2009 ASHRAE Handbook-Fundamental 30.34 1. (30%) As a part of the liquefaction process ammonia goes through an ideal compressor and a heat exchanger. After that supercritical ammonia goes through a throttling valve. Ammonia en ters the compressor at T₁ = 60°C and P₁ = 20 bar and is compressed to the pressure P₁ = 200 bar, and then cooled down in the heat exchanger to T3 = 140°C and flashed at the pressure P = 1 bar Use the attached PH diagram for ammonia to answer the following questions: A. (10) Draw this process on the attached PH diagram. B. (5) Calculate the fraction of ammonia liquefied. C. (5) Calculate the work performed in the compressor. D. (5) Calculate the amount of heat rejected in the heat exchanger. E. (5) How would the fraction of ammonia liquefied change if the amount of heat rejected in the heat exchanger remained the same, but the compressor is replaced with a non-ideal one? (Briefly explain). PRESSURE, MPa -200 40 20 10 8 6 4 2 1864 0.8 0.6 0.2 0.1 0.08 0.06 0.04 0.01 0.02 -200 60 0 0 R-717 (ammonia) Reference state h=200.0 kkg. = 1.00 kinkg-K] for saturated liquid et 0°C 0.40 200 Properties computed with NIST REFPROP O PONA 200 OB 400 20 400 600 8 8 600 500 800 15 6 40 800 450 1000 400 140 T= 20°C 250 60 ep 120 EL 100 1200 300 9 1200 3 100 1000 ENTHALPY, kJ/kg 250 1400 1400 200 1600 1600 150 1800 1800 8.00- 2000 p=100 kg/m³ 80 60 2000 4-5 = 8.40 kJ/(kg K) 40 8.80 2200 30 20 15 10 8.0 6.0 4.0 3.0 2.0 15 1.0 0.80 0.60 2200 0.40 0:30 0.20 0.15 0.10 0.080 0.060 2400 10 8 6 40 20 4 2 2400 1 0.8 06 0.2 0.08 0.06 0.04 0.01 0.02 and on fostulation af Tile Roth (1203 2009 ASHRAE Handbook-Fundamental 30.34
Expert Answer:
Answer rating: 100% (QA)
A The process can be drawn on the PH diagram as follows B The fraction of ammonia liquefied can be c... View the full answer

Related Book For 

Fundamentals of Cost Accounting
ISBN: 978-1259565403
5th edition
Authors: William Lanen, Shannon Anderson, Michael Maher
Posted Date:
Students also viewed these mechanical engineering questions
-
As a part of an employment interview, you are given the partial income statement and selected financial ratios shown for Sneaky Pete's, a chain of western stores. Sneaky Pete's is organized into two...
-
What is the maximum number of stereoisomers possible for the following compound? Br HO >
-
As a part of their customer-service program, United Airlines randomly selected 10 passengers from todays 9 a.m. ChicagoTampa flight. Each sampled passenger is to be inter-viewed in depth regarding...
-
During a worldwide recession in 1983, the oil cartel began to lower prices. Why would a recession make the cartel more vulnerable to price cutting? How would the reduced demand be shared between the...
-
Water at 1000 kPa, 25oC enters a 1-m-diameter horizontal pipe with a steady velocity of 10 m/s. At the exit the pressure drops to 950 kPa due to viscous resistance. Assuming steady state flow,...
-
For a private company client that follows GAAP, auditors must consider the going concern assumption for a reasonable period of time, which is: a. One year from the date the financial statements are...
-
If \(\mathbf{Z}\) is the \(n \times k\) matrix of standardized regressors and \(\mathbf{T}\) is the \(k \times k\) upper triangular matrix in Eq. (11.3), show that the transformed regressors...
-
Financial statements for Allendale Company follow: ALLENDALE COMPANY Balance Sheets As of December 31 2019 2018 Assets Current assets Cash $ 40,000 $ 36,000 Marketable securities 20,000 6,000...
-
Concord Corporation began operations in July 2022. At the end of the month, the company prepares monthly financial statements. It has the following information for the month. 1. At July 31, the...
-
The following cross tabulation shows the average speed of the 25 winners by year of the Daytona 500 automobile race (The 2013 World Almanac). a. Calculate the row percentages. b. W hat is the...
-
The table below shows the responses for two of the variables from the STAT101 Survey in Week 1. We asked you, among other questions, "Which of the choices below best describes your major for your...
-
Lynn Goldsmith is a photographer known for her photographs of famous musicians. In 1981, Goldsmith had a photography session with the singer Prince. Three years later, Vanity Fair obtained a license...
-
A contract is created to refurbish a luxury yacht: new color schemes, new furniture, new wall and floor coverings, new light fixtures, and window treatmentsthe whole works. Of course, it is not just...
-
On February 20, 2009, Cedar Valley Aviation, a wholly owned subsidiary of Aerial Services, Inc. (ASI), brought a Piper 522AS (Cheyenne II) in for maintenance to Des Moines Flying Service, Inc....
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
Should the requirements of the UCC be subject to the application of reliance theories? Go back and review the facts in Case 21-3 about the coal contract. Should silence followed by contract execution...
-
There are several different formulas routinely used to calculate interest. Write a program that lets the user compare three different interest formulas. You will use the simple, compound, and the...
-
Transform the while loop from the previous exercise into an equivalent for loop (make sure it produces the same output).
-
When preparing cost estimates for account analysis purposes, should the costs be extracted from the historical accounting records?
-
A large company has hired your friend. She confides in you about a problem with her boss. Her boss has asked customers to sign sales agreements just before the end of the year, indicating a sale has...
-
There is no reason to investigate favorable variances; only unfavorable variances indicate problems. Do you agree?
-
Among your colleagues in class, identify a term or phrase italicized in this chapter that you think is the most significant from your reading. Absent team consensus, then just provide your...
-
What are the two key financial objectives in the management of a company? How can a focus on these objectives create ethical dilemmas?
-
What is the purpose of financial statements? Would you want to produce them even if they were not required, say, for entity tax reporting?

Study smarter with the SolutionInn App


