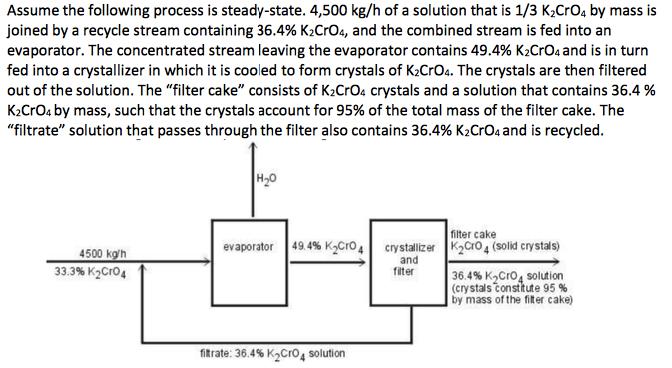
Assume the following process is steady-state. 4,500 kg/h of a solution that is 1/3 K2CrO4 by...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Assume the following process is steady-state. 4,500 kg/h of a solution that is 1/3 K2CrO4 by mass is joined by a recycle stream containing 36.4% K2CrO4, and the combined stream is fed into an evaporator. The concentrated stream leaving the evaporator contains 49.4% K2CrOa and is in turn fed into a crystallizer in which it is cooled to form crystals of K2CrO4. The crystals are then filtered out of the solution. The "filter cake" consists of K2CrO4 crystals and a solution that contains 36.4 % K2CrOa by mass, such that the crystals account for 95% of the total mass of the filter cake. The "filtrate" solution that passes through the filter also contains 36.4% K2CrO4 and is recycled. |н-о filter cake crystallizer K2Cro4 (solid crystals) evaporator 49.4% KCro4 4500 kg'h 33.3% K2Cro4 and filter 36.4% K,Cro, solution (crystals tonstkute 95 % by mass of the fiter cake) fitrate: 36.4% K2Cro4 solution a. What are: (i) mass rate of evaporation, (ii) the mass rate of production of K2CrO, crystals, (iii) the mass feed rates that the evaporator and the crystallizer must handle, and (iv) the recycle ratio? b. What would be the mass production rate of the crystals if the filtrate was discarded instead of recycled? Assume the following process is steady-state. 4,500 kg/h of a solution that is 1/3 K2CrO4 by mass is joined by a recycle stream containing 36.4% K2CrO4, and the combined stream is fed into an evaporator. The concentrated stream leaving the evaporator contains 49.4% K2CrOa and is in turn fed into a crystallizer in which it is cooled to form crystals of K2CrO4. The crystals are then filtered out of the solution. The "filter cake" consists of K2CrO4 crystals and a solution that contains 36.4 % K2CrOa by mass, such that the crystals account for 95% of the total mass of the filter cake. The "filtrate" solution that passes through the filter also contains 36.4% K2CrO4 and is recycled. |н-о filter cake crystallizer K2Cro4 (solid crystals) evaporator 49.4% KCro4 4500 kg'h 33.3% K2Cro4 and filter 36.4% K,Cro, solution (crystals tonstkute 95 % by mass of the fiter cake) fitrate: 36.4% K2Cro4 solution a. What are: (i) mass rate of evaporation, (ii) the mass rate of production of K2CrO, crystals, (iii) the mass feed rates that the evaporator and the crystallizer must handle, and (iv) the recycle ratio? b. What would be the mass production rate of the crystals if the filtrate was discarded instead of recycled?
Expert Answer:
Answer rating: 100% (QA)
Thus iRate of evaporation Qw 2952 kgh ii Rate of produ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A 250-mL portion of a solution that contains 1.5 mM Cu(NO 3 ) 2 is mixed with a solution that is 0.100 M NaCN. After equilibrium is reached, what concentration of Cu 2 + (aq) remains?
-
The following process is used by the Otis Company to process cash receipts. Describe the positive and negative elements of this process. Include operational, information, and control issues. What...
-
(a) Calculate the molarity of a solution that contains 0.175 mol ZnCl2 in exactly 150 mL of solution. (b) How many moles of HCl are present in 35.0 mL of a 4.50 M solution of nitric acid? (c) How...
-
e) Based on your investigation of the data set create two well-formatted tables that illustrate some important features of the data. Your tables should have informative labels and a readable number...
-
Let Y(t) denote the random process corresponding to the transmission of one symbol over the QPSK communications system of Example 13.6. What is the sample space of the underlying experiment? Sketch...
-
Do middle-class heads of households who have a high school diploma only have more assets (ASSET) than those who did not finish high school (EDCL: 1 = No high school diploma, 2 = High school diploma)?...
-
An often-ignored concept in breach of contract is the availability, if any, of the award of punitive damages. Often, cases incorporate both breach of contract and tort actions. The tort actions...
-
The following transactions were completed by The Irvine Company during the current fiscal year ended December 31: Feb. 8. Received 40% of the $18,000 balance owed by DeCoy Co., a bankrupt business,...
-
asap please 1. Determine a maintenance priority by calculating Preventive Maintenance Factors (PMFs) for the three pieces of equipment listed in the table below. Clearly state which piece of...
-
This program currently fails to compile. Modify TripleMin() so that item1 can be of a different type than item2 and item3. #include <iostream> using namespace std; template<typename...
-
Talk with three people to get their opinions on federal spending on energy, education, and defense. You may ask friends, family members, and other trusted adults. Ask them to rank the programs listed...
-
What is the main purpose of the Organization of the Petroleum Exporting Countries (OPEC)? A. Regulating international trade B. Promoting renewable energy sources C. Controlling the production and...
-
A dynamic system has the following transfer function. What is the unit step response? Use graphical methods to compute partial fraction expansion. H(s)= 100(s+2) (s+10) (s+5s+49)
-
According to FASB, when should a company journalize a contingent liability? ... O A. Journalize the contingent liability, even though you will probably win the lawsuit. B. Journalize the contingent...
-
Please view this Ted Talk : https://www.ted.com/talks/derek_sivers_weird_or_just_different?language=en 1. Have you ever felt like a stranger or a minority? If so, how did that feel? 2. Have you ever...
-
What is the correct Cost of Goods Sold under the FIFO method given the following information? Beginning Inventory: 50 units @10, Purchase 1: 15 units @13, Purchase 2: 15 units @15, Ending Inventory:...
-
Analyse base case (expected) cash flows and their potential uncertainty. For your base case analysis, calculate the five investment decision criteria NPV, IRR, PI, payback period, discounted payback...
-
Write a paper about medication error system 2016.
-
Consider the following equilibrium process at 686°C: The equilibrium concentrations of the reacting species are [CO] = 0.050 M, [H2] = 0.045 M, [CO2] = 0.086 M, and [H2O] = 0.040 M. (a) Calculate...
-
Many hydrocarbons exist as structural isomers, which are compounds that have the same molecular formula but different structures. For example, both butane and isobutane have the same molecular...
-
Draw orbital diagrams for atoms with the following electron configurations: (a) 1s22s22p5 (b) 1s22s22p63s23p3 (c) 1s22s22p63s23p64s23d7
-
Discuss the importance of validity and research design. Next, choose one type of validity (internal, external, construct, or statistical conclusion) and discuss its relevance to experimental, quasi...
-
Explain from a technical viewpoint why it is important to distinguish a method, research, approach, and design. Next, briefly discuss how understanding each term individually in addition to how these...
-
Define the two major types of sampling strategies .

Study smarter with the SolutionInn App


