Astrik Ltd anticipates the following will occur in February. 1. Sales are expected to be $420...
Fantastic news! We've Found the answer you've been seeking!
Question:
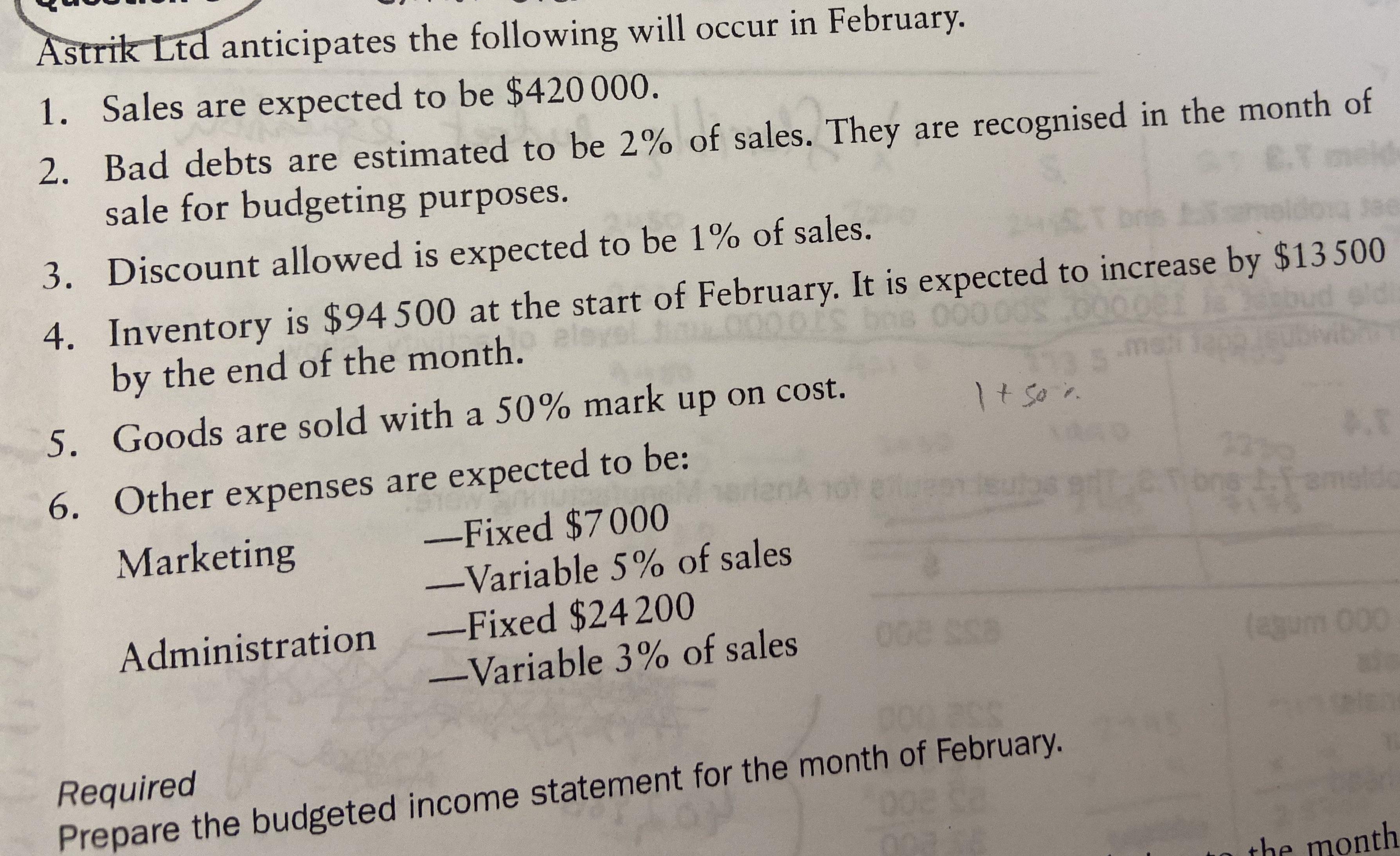
Transcribed Image Text:
Astrik Ltd anticipates the following will occur in February. 1. Sales are expected to be $420 000. 2. Bad debts are estimated to be 2% of sales. They are recognised in the month of sale for budgeting purposes. E.T meid 3. Discount allowed is expected to be 1% of sales. LT bris Sm long Jae 000 00s 2000 4. Inventory is $94 500 at the start of February. It is expected to increase by $13500 by the end of the month. 5. Goods are sold with a 50% mark up on cost. 6. Other expenses are expected to be: enA Marketing -Fixed $7000 -Variable 5% of sales Administration -Fixed $24 200 -Variable 3% of sales 1+ 50%. 008 308 Required Prepare the budgeted income statement for the month of February. mah ns1.1-3 amaldo (agum 000 the month Astrik Ltd anticipates the following will occur in February. 1. Sales are expected to be $420 000. 2. Bad debts are estimated to be 2% of sales. They are recognised in the month of sale for budgeting purposes. E.T meid 3. Discount allowed is expected to be 1% of sales. LT bris Sm long Jae 000 00s 2000 4. Inventory is $94 500 at the start of February. It is expected to increase by $13500 by the end of the month. 5. Goods are sold with a 50% mark up on cost. 6. Other expenses are expected to be: enA Marketing -Fixed $7000 -Variable 5% of sales Administration -Fixed $24 200 -Variable 3% of sales 1+ 50%. 008 308 Required Prepare the budgeted income statement for the month of February. mah ns1.1-3 amaldo (agum 000 the month
Expert Answer:
Answer rating: 100% (QA)
ASTRIK LTD Budgeted income statement for the month of Febr... View the full answer

Related Book For 

Fundamentals of Financial Management
ISBN: 978-0324597707
12th edition
Authors: Eugene F. Brigham, Joel F. Houston
Posted Date:
Students also viewed these accounting questions
-
The Thompson Toy Company manufactures toy building block sets for children. Thompson is planning for 2017 by developing a master budget by quarters. Thompsons balance sheet for December 31, 2016,...
-
The Grady Tire Company manufactures racing tires for bicycles. Grady sells tires for $60 each. Grady is planning for the next year by developing a master budget by quarters. Gradys balance sheet for...
-
The Grilton Tire Company manufactures racing tires for bicycles. Grilton sells tires for $50 each. Grilton is planning for the next year by developing a master budget by quarters. Griltons balance...
-
Use the figure to make the lists in Exercises 8586. List the slopes m 1 , m 2 , m 3 , and m 4 in order of decreasing size. y y = mx + b y = mx + b X - y = m3x + b3 y = m4x + b4
-
One of the biggest problems of student writers is paraphrasing secondary sources correctly to avoid plagiarism. Your Task. For each of the following, read the original passage. Analyze the...
-
For the circuit in Fig. 3.123 , find the gain v o v s . 3.5 k2 500 2 ww- Vo 80 v, 500 2 Vs 500 2
-
The internal energy of an ideal gas depends on the (a) Pressure and temperature (b) Volume and temperature (c) Volume and pressure (d) Temperature only.
-
1. In what way(s) has Ken Frazier demonstrated his effectiveness as a crisis leader? 2. Given our discussion on the distinction between a traditional and learning organization, what type of...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...
-
The shareholders equity accounts of Tmao Inc. at December 31, 2023, are as follows: Preferred shares, $3 noncumulative, unlimited number authorized, 4,000 issued ......... $400,000 Common shares,...
-
DETERMINE THE STAGE OF PRODUCT LIFE CYCLE. Price reductions are used as a tool of competition. a . Introduction stage b . Growth stage c . Maturity stage d . Decline stage.
-
A stream of reactant A (C A0 = 3, C RO = 0, C CO = 0) is to be decomposed by the following microbial fermentation What is the lowest C A which can be obtained in a single mixed flow reactor of size V...
-
Evaluate the extent to which the proposed changes to IAS 1 are likely improve the decision-usefulness of published financial statements for users.
-
Which statement is false? a) The poor pay higher prices to buy groceries, furniture, and appliances. b) Low-income families can pay over $500 more for the same car bought by a higher-income...
-
Which one of the following has been the least effective antipoverty program _______? a) the Welfare Reform Act of 1996 b) Social Security c) food stamps d) the Earned Income Tax Credit
-
Which statement is the most accurate? a) The welfare rolls today are much lower than they were in 1996. b) About one in nine Americans lives below the poverty line. c) Without Social Security...
-
Suppose that the tax rate for the company is 40%. The company has both financial expenses and financial income. Financial expenses are 40O TL. What should be the financial income? 725 TL 625 TL 375...
-
Define the essential properties of the following types of operating systems: a. Batch b. Interactive c. Time sharing d. Real time e. Network f. Parallel g. Distributed h. Clustered i. Handheld
-
Assume that the risk-free rate is 5% and the market risk premium is 6%. What is the expected return for the overall stock market? What is the required rate of return on a stock with a beta of 1.2?
-
What is a post-audit, why do firms use them, and what problems can arise when they are used?
-
The Howe Computer Company has grown rapidly during the past 5 years. Recently, its commercial bank urged the company to consider increasing its permanent financing. Its bank loan under a line of...
-
Calculate the entropy change when \(96 \mathrm{~g}\) of methane is heated from \(35^{\circ} \mathrm{C}\) to \(200^{\circ} \mathrm{C}\) at constant volume. Assume \(C_{V}=1.735 \mathrm{~kJ} /...
-
\(5.105 \mathrm{~mol}\) of an ideal gas is expanded from a temperature of \(300 \mathrm{~K}\) at 3 bars to \(400 \mathrm{~K}\) at 12 bars. Assume that \(C_{P}=26.73 \mathrm{~kJ} /...
-
Calculate \(\Delta S\) when \(8 \mathrm{~mol}\) of an ideal gas are heated from a temperature of 350 \(\mathrm{K}\) to a temperature of \(700 \mathrm{~K}\) at constant pressure. Assume that...

Study smarter with the SolutionInn App


