At 1 atm, how much energy is required to heat 89.0 g of HO(s) at-16.0 C...
Fantastic news! We've Found the answer you've been seeking!
Question:
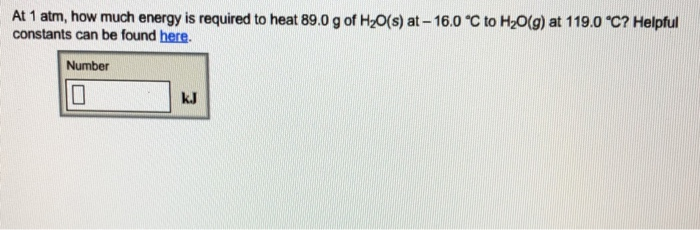

Transcribed Image Text:
At 1 atm, how much energy is required to heat 89.0 g of H₂O(s) at-16.0 °C to H₂O(g) at 119.0 °C? Helpful constants can be found here. Number kJ Heat-transfer constants for H₂0 at 1 atm Quantity Enthalpy of fusion Enthalpy of vaporization Specific heat of solid H₂O (ice) 2.087 J/(g-°C) * Specific heat of liquid H₂O (water) 4.184 J/(g-°C)* Specific heat of gaseous H₂O (steam) 2.000 J/(g-°C) * per gram 333.6 J/g 2257 J/g per mole 6010. J/mol 40660 J/mol 37.60 J/(mol-°C) 75.37 0/(mol-°C) 36.03 J/(mol-°C) At 1 atm, how much energy is required to heat 89.0 g of H₂O(s) at-16.0 °C to H₂O(g) at 119.0 °C? Helpful constants can be found here. Number kJ Heat-transfer constants for H₂0 at 1 atm Quantity Enthalpy of fusion Enthalpy of vaporization Specific heat of solid H₂O (ice) 2.087 J/(g-°C) * Specific heat of liquid H₂O (water) 4.184 J/(g-°C)* Specific heat of gaseous H₂O (steam) 2.000 J/(g-°C) * per gram 333.6 J/g 2257 J/g per mole 6010. J/mol 40660 J/mol 37.60 J/(mol-°C) 75.37 0/(mol-°C) 36.03 J/(mol-°C)
Expert Answer:
Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these chemistry questions
-
How much energy is required to remove? (a) A proton. (b) A neutron, from 16/8O? Explain the difference in you answers.
-
How much energy is required to produce a neutron anti neutron pair?
-
How much energy is required to remove an electron from a hydrogen atom in the n = 4 state?
-
List and describe the payment options (terms of sale) that can be applied to domestic U.S. shipments.
-
You are a team leader of four systems analysts. You have one analyst who has never done a structured walkthrough of her work. How would you help the analyst to get started? How would you ensure that...
-
The unadjusted trial balance of Oak and Brass Interiors at December 31, 2012, the end of the current year, is shown below. The data needed to determine year-end adjustments are as follows:a. Supplies...
-
Superhero Industries reported the following at September 30: Requirements 1. Record retirement of half of the bonds on October 1 at the call price of 101. 2. Record conversion of the remainder of the...
-
Listed below are several terms and phrases associated with the FASBs conceptual framework. Pair each item from List A (by letter) with the item from List B that is most appropriately associated with...
-
You are the financial manager of Ferry Constructions, a small construction firm. The owners are concerned about their inventory management, where up until now, ordering of raw materials has been...
-
Explain the difference between a discrete and a continuous variable. Give an example of each not included in the text.
-
The management of NT Airways is thinking about introducing a daily return-flight from Darwin to Singapore. You are the management accountant at NT Airways and are working with the marketing manager...
-
An ingot of germanium is formed by melting together 1 kg of germanium and 118 g of arsenic. (i) Assuming that the arsenic is uniformly distributed, calculate the number of arsenic atoms per unit of...
-
Question: Assume that Trump acquired Clinton on January 2, 2001 Trump issued 60,000 new shares of its common stock valued at $4.00 per share for all of the outstanding stock of Clinton. Immediately...
-
How do modern operating systems implement secure boot mechanisms, and what role does hardware-based security play in this process ? Explain
-
Analyze and differentiate between ADA and CAATTS tools when assessing internal controls, audit risk, and gathering substantive evidence. Explain the potential application of ADA in evaluating audit...
-
Question: 10/25/YR1-- YSR Inc. acquires supplies to be used by its employees. The supplies cost $2,000. Although YSR Inc. has received the supplies, it will not pay for the supplies until November,...
-
Develop code that allows a user to log in. This means you should focus on the following functionality: The app should check the username and password against the database when the user attempts to...
-
Before the latest financial crisis and recession, when was the largest recession of the past 50 years, and what was the cumulative loss in output over the course of the slowdown?
-
A firecracker is tossed straight up into the air. It explodes into three pieces of equal mass just as it reaches the highest point. Two pieces move off at 120 m/s at right angles to each other. How...
-
The nucleus in a 127N atom captures one of the atom's electrons, changing the nucleus to 126C and emitting a neutrino. What is the total energy of the emitted neutrino?
-
The currents in household wiring and power lines alternate at a frequency of 60.0 Hz. (a) What is the wavelength of the EM waves emitted by the wiring? (b) Compare this wavelength with Earth's...
-
a. Nonlinearity in mass b. Nonlinearity in damping c. Linear equation d. Nonlinearity in spring \(\ddot{x}+f \frac{\dot{x}}{|\dot{x}|}+\omega_{n}^{2} x=0\)
-
The equation of motion of a nonlinear system is given by \[\ddot{x}+c \dot{x}+k_{1} x+k_{2} x^{2}=a \cos 2 \omega t\] Investigate the subharmonic solution of order 2 for this system.
-
Prove that, for the system considered in Section 13.5.1, the minimum value of \(\omega^{2}\) for which the amplitude of subharmonic oscillations \(A\) will have a real value is given by...

Study smarter with the SolutionInn App



