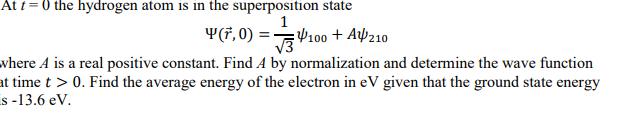At t =0 the hydrogen atom is in the superposition state 1 Y(F, 0) V100 +...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
At t =0 the hydrogen atom is in the superposition state 1 Y(F, 0) V100 + A210 where A is a real positive constant. Find A by normalization and determine the wave function at time t > 0. Find the average energy of the electron in eV given that the ground state energy s -13.6 eV. At t =0 the hydrogen atom is in the superposition state 1 Y(F, 0) V100 + A210 where A is a real positive constant. Find A by normalization and determine the wave function at time t > 0. Find the average energy of the electron in eV given that the ground state energy s -13.6 eV.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
An electron in a hydrogen atom is in a state with l = 5.What is the minimum possible value of the semi-classical angle between and Lz?
-
The hydrogen atom is composed of one proton in the nucleus and one electron, which moves about the nucleus. In the quantum theory of atomic structure, it is assumed that the electron does not move in...
-
Consider a transition in which the hydrogen atom is excited from n = 1 to n = . (a) What is the end result of this transition? (b) What is the wavelength of light that must be absorbed to accomplish...
-
A close company which prepares accounts to 31 March each year is owned and managed by a single shareholder/director who is not a Scottish taxpayer and who is paid a salary of 5,000 per month. In...
-
The C NMR spectrum of 2-ethylbutanal consists of the following absorptions: 11.5, 21.7, 55.2, and 204.7. Draw the structure of this aldehyde, label each chemically non-equivalent set of carbons,...
-
All companies strive to make a profit. Breaking even every year is not considered an acceptable return on an organization's investment. Explain why, in spite of these statements, it is still crucial...
-
A study was conducted to investigate the relationship between severe headaches and being left- or right-handed. (Incidentally, Lisa Kudrow, who played Phoebe Buffay on the hit sitcom "Friends," is an...
-
Siena Industries (a sole proprietorship) sold three 1231 assets on October 10, 2014. Data on these property dispositions are as follows. a. Determine the amount and the character of the recognized...
-
Your company needs fresh cash (10 Mio ). In the board meeting the CFO was proposing 10,000 bonds with yield to maturity of 100 % for a) 1 or b)2 years with face values of 1,000 and coupons of i) 10...
-
Paolo is a 50% partner in the Capri Partnership and has decided to terminate his partnership interest. Paolo is considering two options as potential exit strategies. The first is to sell his...
-
How much energy required to heat the internal volume of the pot from 15C to 1650C. Dimensions: 2m diameter Stainless steel 316 5mm thick 2m high a. Burner will be firing inside the pot heating the...
-
A 2 5 . 4 g piece of iron and a 2 4 . 2 g piece of gold at 1 0 0 . 0 degrees celsius were dropped into 6 1 0 . 0 mL of water at 2 7 . 0 degrees Celsius. The molar heat capacities of iron and gold are...
-
What's the partial pressure of carbon dioxide in a container that holds 5 moles of carbon dioxide, 3 moles of nitrogen, and 1 mole of hydrogen and has a total pressure of 1.05 atm?
-
What is the molar solubility ( in terms of mol / L ) of SrF 2 in pure water? Note the Ksp of SrF 2 is 2 . 6 x 1 0 - 1 0 Molar solubility of SrF 2 = 1 . 9 x 1 0 - 3 MMolar solubility of SrF 2 = 8 . 7...
-
Write the net ionic equation for the equilibrium that is established when ammonium iodide is dissolved in water. (Use H3O+ instead of H+. It is not necessary to include states such as (aq) or (!).) +...
-
You measure the amount of carbon-14 (the parent isotope) and nitrogen-14 (the daughter isotope) in a sample of bone and determine that 0.75 half-lives have elapsed for carbon-14. What is the age of...
-
Albert who is a resident of Canada attended the University of Toronto on a full-time basis for 8 months during 2023. While there he earned employment income for tax purposes of $19,000 for 2023. He...
-
What is the ideal number of children to have? This question was asked on the Sullivan Statistics Survey I. Draw a dot plot of the variable Children from theSullivanStatsSurveyI data set at...
-
Solve the differential equation y' = x + y by making the change of variable u = x + y.
-
A cross-section of an airplane wing is shown. Measurements of the thickness of the wing, in centimeters, at 20-centimeter intervals are, 5.8, 20.3, 26.7, 29.0, 27.6, 27.3, 23.8, 20.5, 15.1, 8.7, and...
-
Under ideal conditions a certain bacteria population is known to double every three hours. Suppose that there are initially 100 bacteria. (a) What is the size of the population after 15 hours? (b)...
-
LDDS continued to publicly report increasing profits and sales in the financial statements, which allowed it to acquire more companies with no limit to the growth of its stock price. True/False
-
LDDS suffered from high fixed costs coupled with a lack of technical knowledge and expertise in optimizing the configuration of circuits, which resulted in high line costs. True/False
-
WorldCom overstated its sales by holding its books open at the close of a reporting period. True/False

Study smarter with the SolutionInn App