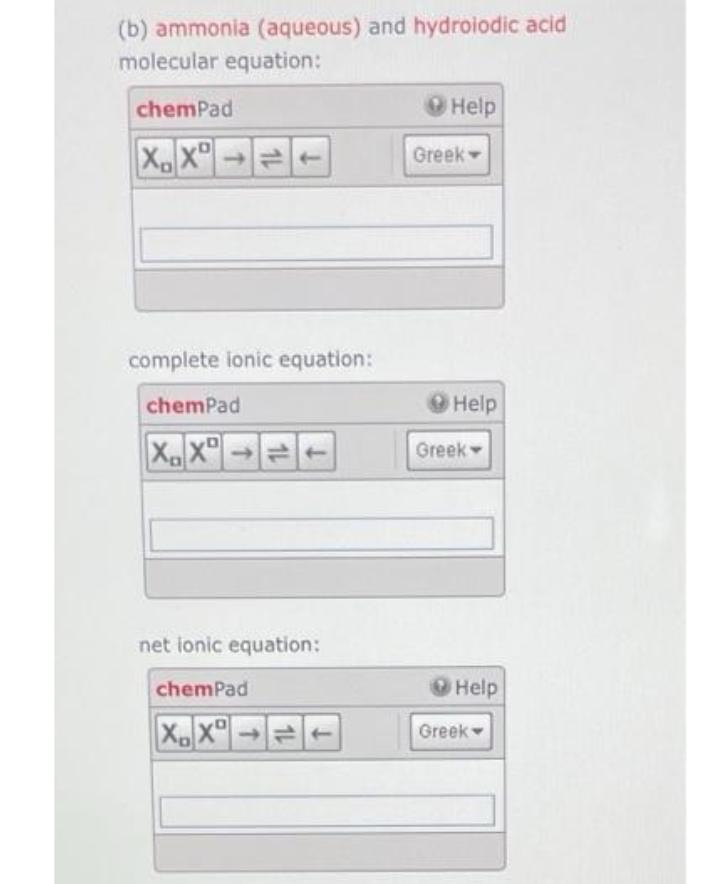
(b) ammonia (aqueous) and hydroiodic acid molecular equation: chemPad XXT complete ionic equation: chemPad XDXD net...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
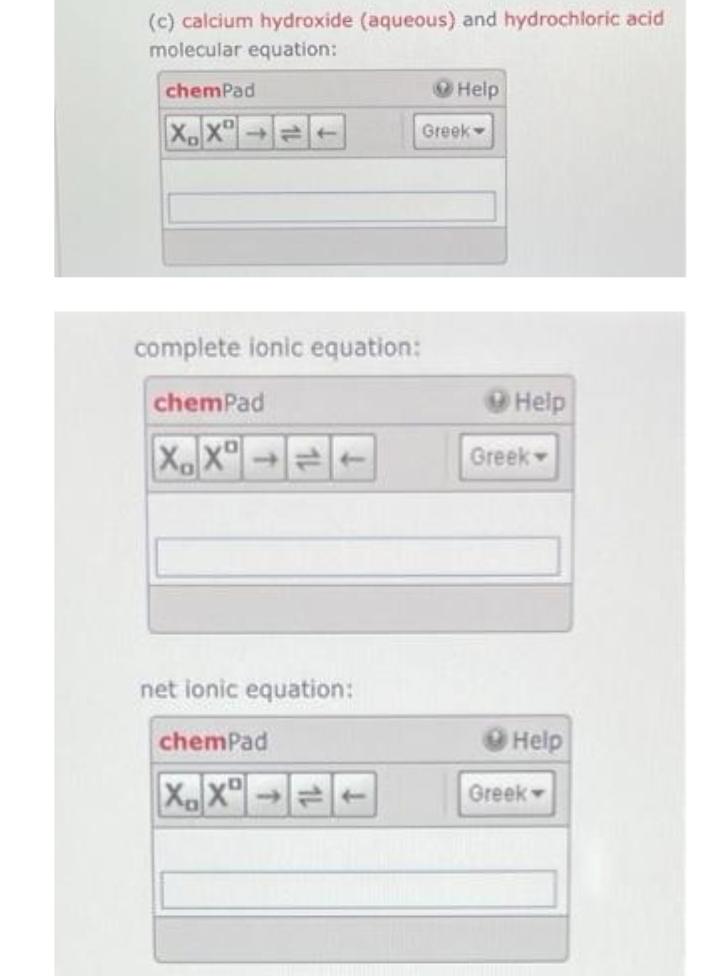
(b) ammonia (aqueous) and hydroiodic acid molecular equation: chemPad XX→T complete ionic equation: chemPad XDXD net ionic equation: chemPad XX Help Greek- Help Greek Help Greek: (c) calcium hydroxide (aqueous) and hydrochloric acid molecular equation: chemPad XDXD 1 complete ionic equation: chemPad XX net ionic equation: chemPad XX→= Help Greek- Help Greek Help Greek- (b) ammonia (aqueous) and hydroiodic acid molecular equation: chemPad XX→T complete ionic equation: chemPad XDXD net ionic equation: chemPad XX Help Greek- Help Greek Help Greek: (c) calcium hydroxide (aqueous) and hydrochloric acid molecular equation: chemPad XDXD 1 complete ionic equation: chemPad XX net ionic equation: chemPad XX→= Help Greek- Help Greek Help Greek-
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Complete and balance the following molecular equations, and then write the net ionic equation for each: (a) HBr(aq) + Ca(OH)2(aq) (b) Cu (OH)2 (s) + HCIO4 (aq) (c) Al(OH)3(s) + HNO3(aq)
-
a. The equation for the reaction of strontium hydroxide with hydrochloric acid is shown below. Sr(OH) 2 + 2HCl SrCl 2 + 2H 2 O 25.0 cm 3 of a solution of strontium hydroxide was exactly neutralised...
-
Solid sodium carbonate reacts with aqueous hydrochloric acid to form aqueous sodium chloride, carbon dioxide and water. Na 2 CO 3 + 2HCl 2NaCl + CO 2 + H 2 O a. Rewrite this equation to include...
-
During one week, four production employees of Martinez Manufacturing Company worked the hours shown below. All these employees receive overtime pay at one and one-half times their regular hourly rate...
-
Prepare a depreciation schedule to be used for tax purposes for $40,000 of computer equipment using the 150% declining-balance method and a half-year convention. Ignore any special depreciation...
-
What is the name of the set of microtubules that pull chromosomes apart? What is the name of the attachment point between microtubules and chromosomes? What is the name of the ends of the cells where...
-
Dafna Brauning owns a small chain of fast-casual restaurants in Denver that sells sandwiches, soups, wraps, and desserts. In general, these restaurants are successful, but Dafna feels that they are...
-
Canned fruits and vegetables are the main products made by Good Foods, Inc. All direct materials are added at the beginning of the Mixing Department's process. When the ingredients have been mixed,...
-
a) Explain the following accounting concepts: i. Business entity Revenue realization ii. Accruals iii. iv. V. Substance over form Conservatism vi. Going concern (6 Marks) b) Classify the following...
-
All cash flows occur at the end of the period. 1. What is the annual interest rate, if $1,000 grows to $1,338 over 5 years? (4) remember to use opposite signs on the payment and present values 2....
-
What characteristics make a law "quasi constitutional"? What are the effects of characterizing a law as quasi-constitutional? What are the typical grounds upon which human rights codes prohibit...
-
Many years ago, test pilots accelerating through Mach 1 reported that the ride was pretty rough just before Mach 1 but smoothed out suddenly as Mach 1 was breached. How can you account for this?
-
Is it possible for the same medium to carry both a longitudinal and a transverse wave? If so, give an example. If not, explain why not.
-
Electric eels live in fresh water. The torpedo ray is an electric fish that lives in salt water. The electrocytes in the ray are grouped differently than in the eel; each stack of electrocytes has...
-
Suppose you have a light spring stretched out and one end is attached to a wall. With this setup, you can move the free end in any of three directions \((x, y, z)\). If the spring lies along, say,...
-
While attending an air show, you observe a fighter jet traveling horizontally at a speed of Mach 1. 30. At the instant you hear the sonic boom, what is the angle between your line of sight to the jet...
-
A Master Production Schedule calls for 110 units of Product A. There are currently 60 of Product A on hand. Each A requires 4 of Part B. There are 20 units of B available. The net requirements for B...
-
Show that the block upper triangular matrix A in Example 5 is invertible if and only if both A 11 and A 22 are invertible. Data from in Example 5 EXAMPLE 5 A matrix of the form A = [ A11 A12 0 A22 is...
-
Calculate the pH of a solution made by mixing 0.60 L of 0.10 M NH4Cl with 0.50 L of 0.10 M NaOH. Kb for NH3 is 1.8 10-5.
-
Name the following molecular compounds. a. AsBr3 b. H2Se c. P2O5 d. SiO2
-
Calculate the pH of a solution obtained by mixing 500.0 mL of 0.10 M NH3 with 200.0 mL of 0.15 M HCl.
-
Light of a single wavelength is incident on a diffraction grating with \(500 \mathrm{slits} / \mathrm{mm}\). Several bright fringes are observed on a screen behind the grating, including one at...
-
The shiny surface of a CD is imprinted with millions of tiny pits, arranged in a pattern of thousands of essentially concentric circles that act like a reflection grating when light shines on them....
-
If sunlight shines straight onto a peacock feather, the feather appears bright blue when viewed from \(15^{\circ}\) on either side of the incident beam of sunlight. The blue color is due to...

Study smarter with the SolutionInn App



