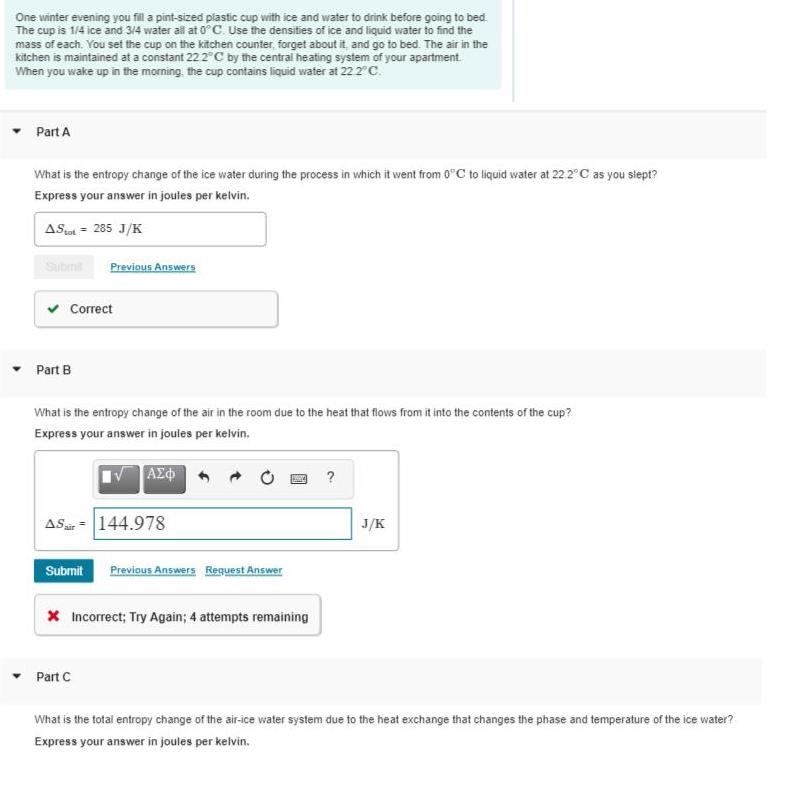
One winter evening you fill a pint-sized plastic cup with ice and water to drink before...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
One winter evening you fill a pint-sized plastic cup with ice and water to drink before going to bed. The cup is 1/4 ice and 3/4 water all at 0° Cc. Use the densities of ice and liquid water to find the mass of each. You set the cup on the kitchen counter, forget about it, and go to bed. The air in the kitchen is maintained at a constant 22 2"C by the central heating system of your apartment When you wake up in the morning, the cup contains liquid water at 22 2°C. Part A What is the entropy change of the ice water during the process in which it went from 0°C to liquid water at 22.2"C as you slept? Express your answer in jouies per kelvin. ASat = 285 J/K Subit Previous Answers v Correct Part B What is the entropy change of the air in the room due to the heat that flows from it into the contents of the cup? Express your answer in joules per kelvin, ? ASa = 144.978 J/K Submit Previous Answers Request Answer X Incorrect; Try Again; 4 attempts remaining Part C What is the total entropy change of the air-ice water system due to the heat exchange that changes the phase and temperature of the ice water? Express your answer in joules per kelvin. One winter evening you fill a pint-sized plastic cup with ice and water to drink before going to bed. The cup is 1/4 ice and 3/4 water all at 0° Cc. Use the densities of ice and liquid water to find the mass of each. You set the cup on the kitchen counter, forget about it, and go to bed. The air in the kitchen is maintained at a constant 22 2"C by the central heating system of your apartment When you wake up in the morning, the cup contains liquid water at 22 2°C. Part A What is the entropy change of the ice water during the process in which it went from 0°C to liquid water at 22.2"C as you slept? Express your answer in jouies per kelvin. ASat = 285 J/K Subit Previous Answers v Correct Part B What is the entropy change of the air in the room due to the heat that flows from it into the contents of the cup? Express your answer in joules per kelvin, ? ASa = 144.978 J/K Submit Previous Answers Request Answer X Incorrect; Try Again; 4 attempts remaining Part C What is the total entropy change of the air-ice water system due to the heat exchange that changes the phase and temperature of the ice water? Express your answer in joules per kelvin.
Expert Answer:
Answer rating: 100% (QA)
Paolin has been solved and correct will solve part and c volume of cup... View the full answer

Posted Date:
Students also viewed these physics questions
-
Air at 600 K flows with 3 kg/s into a heat exchanger and out at 100oC. How much (kg/s) water coming in at 100 kPa, 20oC can the air heat to the boiling point?
-
A room of volume V contains air having equivalent molar mass M (in g/mol). If the temperature of the room is raised from T1 to T2, what mass of air will leave the room? Assume that the air pressure...
-
A counter flowing heat exchanger cools air at 600 K, 400 kPa to 320 K using a supply of water at 20C, 200 kPa. The water flow rate is 0.1 kg/s and the air flow rate is 1 kg/s. Assume this can...
-
Increasingly sophisticated communications technology is allowing more people to work at home. What effect do you think this might have on land-use patterns, specifically the density of residential...
-
You're assistant director of marketing for a maker of upscale furniture, and your company is preparing to enter new markets in California and the Southwest. Entering new markets, especially one of...
-
A large mirror is hung from a nail as shown in Figure. The supporting steel wire has a diameter of 0.2 mm and an unstretched length of 1.7 m. The distance between the points of support at the top of...
-
Chee Yong Chaw began his business on 1 July 2025. The business balances its books at month-end and uses special journals and the periodic inventory system. Transactions for July 2025 were as follows:...
-
Globetron manufactures components for use in small electronic products such as computers, CD players, and radios at plants in Belgium, Germany, and France. The parts are transported by truck to...
-
6. To a ground observer the block C is moving with v, and the block A with v. B is moving with v relative to C as shown in the figure. Identify the correct statement. (a) V-V =Vo (b)= A C B (c) V+V =...
-
Kate Jackson, a new staff accountant, is confused because of the complexities involving accounting standard setting. Specifically, she is confused by the number of bodies issuing financial reporting...
-
A $1,000 face value corporate bond paying 10% is due to expire in 10 years time. The bond is currently selling for $1,064.. You colleague, who has an opportunity cost of 9% yield, is interested in...
-
Outline the advantages and disadvantages of using a DBMS over developing an application using files.
-
What else do we use state machine diagrams for, apart from modelling the state of interface objects?
-
Why is the class constructor private in the Singleton pattern?
-
List in your own words the three steps used in going from an unnormalized relation to a relation in third normal form.
-
Distinguish between task and technique, and give some examples of each.
-
emergency management assistance company is a state to state system for sharing resources during an emergency or disaster true or false
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Financial balances for the car hire business of Terry's Wedding Cars on 31 March 2024 are provided below in a table in accounting equation form similar to the chapter illustrations. During April, the...
-
Trans Clothing Alterations began operations on 1 August 2024 and completed the following transactions during the first month. 1. Tran deposited \($18\) 000 of her personal funds in a current account...
-
Finesse Fitness was established on 1 April 2024 with an initial investment of $60000 by the owner, Daniel Hewitt. During the first few months of business, the owner employed a student studying...

Study smarter with the SolutionInn App

