Question: 1. a. 7P3 3. a. Ps 5. 7. 9. 11. 13. 2. a. gP4 4. a. Po b. 7C3 b. 5C5 . 14P b.
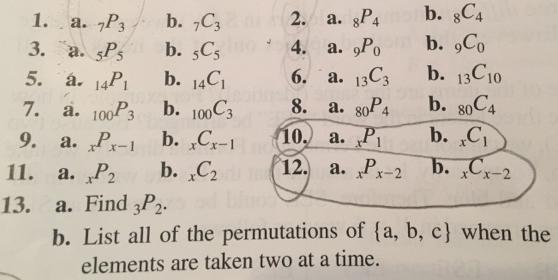
1. a. 7P3 3. a. Ps 5. 7. 9. 11. 13. 2. a. gP4 4. a. Po b. 7C3 b. 5C5 . 14P b. 14C a. 100P3 b. 100 C3 a. P-1 b. Cr-1 a. P2 b. C a. Find 3P2. b. List all of the permutations of (a, b, c) when the elements are taken two at a time. b. 8C4 b. 9Co b. 13C10 b. 80C4 b. C b. C-2 6. a. 13C3 8. a. 80P4 10. a. P 12. a. P-2
Step by Step Solution
3.39 Rating (152 Votes )
There are 3 Steps involved in it
ANSWER 1 a 7P3 represents the number of permutations of 3 objects taken from a set of 7 distinct objects Using the formula for permutations 7P3 7 7 3 7 4 7 6 5 4 4 7 6 5 210 Therefore 7P3 is equal to ... View full answer

Get step-by-step solutions from verified subject matter experts


