Chemical energy is released or absorbed from reactions in various forms. The most easily measurable form...
Fantastic news! We've Found the answer you've been seeking!
Question:
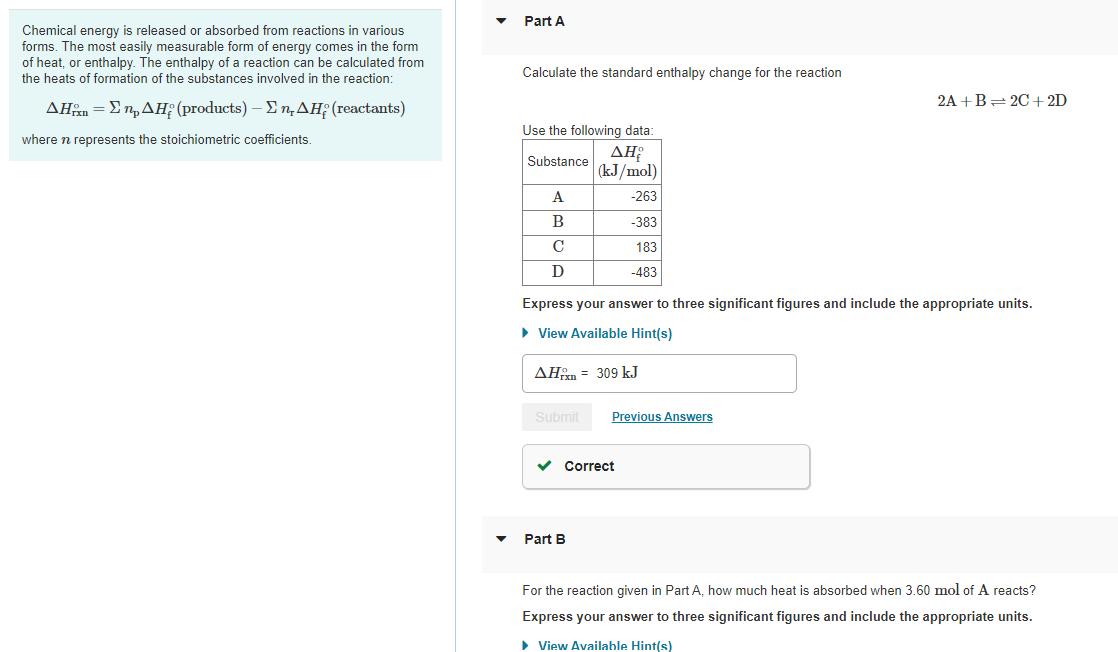
Transcribed Image Text:
Chemical energy is released or absorbed from reactions in various forms. The most easily measurable form of energy comes in the form of heat, or enthalpy. The enthalpy of a reaction can be calculated from the heats of formation of the substances involved in the reaction: ΔΗån = ΣηΔΗ (products) - Ση.ΔΗ (reactants) where n represents the stoichiometric coefficients. ♥ Part A Calculate the standard enthalpy change for the reaction Use the following data: ΔΗ Substance (kJ/mol) A B C D Express your answer to three significant figures and include the appropriate units. ► View Available Hint(s) AH 309 kJ Submit -263 -383 183 -483 ✓ Correct Part B Previous Answers 2A+B=2C+2D For the reaction given in Part A, how much heat is absorbed when 3.60 mol of A reacts? Express your answer to three significant figures and include the appropriate units. ▸ View Available Hint(s) Chemical energy is released or absorbed from reactions in various forms. The most easily measurable form of energy comes in the form of heat, or enthalpy. The enthalpy of a reaction can be calculated from the heats of formation of the substances involved in the reaction: ΔΗån = ΣηΔΗ (products) - Ση.ΔΗ (reactants) where n represents the stoichiometric coefficients. ♥ Part A Calculate the standard enthalpy change for the reaction Use the following data: ΔΗ Substance (kJ/mol) A B C D Express your answer to three significant figures and include the appropriate units. ► View Available Hint(s) AH 309 kJ Submit -263 -383 183 -483 ✓ Correct Part B Previous Answers 2A+B=2C+2D For the reaction given in Part A, how much heat is absorbed when 3.60 mol of A reacts? Express your answer to three significant figures and include the appropriate units. ▸ View Available Hint(s)
Expert Answer:
Related Book For 

Principles of Information Systems
ISBN: 978-0324665284
9th edition
Authors: Ralph M. Stair, George W. Reynolds
Posted Date:
Students also viewed these chemistry questions
-
On the first day of the year (t = 0), a city uses electricity at a rate of 2000 MW. That rate is projected to increase at a rate of 1.3% per year. a. Based on these figures, find an exponential...
-
At what interest rate are these two transactions in Figure P3.8 equivalent? $1.200 $1,000 2 Option I Option 2
-
Your company has been offered credit terms of 4/30, net 90 days. What will be the nominal annual percentage cost of its non free trade credit if it pays 120 days after the purchase? (Assume a 365-day...
-
Assume that investment spending depends only on the interest rate and no longer depends on output. Given this information, a decrease in money supply: a. will cause investment to increase. b. may...
-
Which type of ANOVA is used in a two-way design when (a) Both factors are tested using independent samples? (b) One factor involves independent samples and one factor involves related samples? (c)...
-
Gibbs' free energy of a pure fluid approaches at constant temperature (a) Infinity (b) Minus infinity (c) Zero (d) None of these.
-
Why is a distinction made between conventional accounting and ecological accounting?
-
Acme Parts runs a small factory and employs workers who are paid one of three hourly rates depending on their shift: first shift, $17 per hour; second shift, $18.50 per hour; third shift, $22 per...
-
How does the principle of plasticity influence the design of reinforced concrete structures subjected to complex loading conditions ?
-
The information provided in this test allows you to set up the Cieneral, Payables and Receivables ledgers for an Ontario company named CHRIS'S WELDING owned and run by Chris George, amaster welder....
-
Standard Recipe Card Student ID Name of the dish Dish Description Portion Nos. Portion Size Sales Price Food Cost % Ingredients Item Qty Req. UoM Cost per UoM Yield % Actual Cost Input Calculation...
-
What could be done to get those help and those who have been incarcerated for a long period of time and how would we help them jump back into society? In what ways would we generate job corps...
-
The world long jump record is 9.95 m (Mike Powell, USA, 1991).Treated as a projectile, what is the maximum range, in meters, obtainable by a person if he or she has a take-off speed of 8.9 m/s?...
-
Explain the role of phase equilibrium thermodynamics in the design and optimization of liquid-liquid extraction systems, with a specific emphasis on tie-line data analysis and the prediction of phase...
-
A well-thrown ball is caught in a well-padded mitt.If the deceleration of the ball is 2.25 x 10* m/s?, and 1.9 ms (I ms = 10-3 s) elapses from the time the ball first touches the mitt until it stops,...
-
The tape in a videotape cassette has a total length 278 m and can play for 2.1 h. As the tape starts to play, the full reel has an outer radius of 48 mm and an inner radius of 12 mm. At some point...
-
Find an example of policymaking in the health sector at the US federal level and answer the following questions: a) How is the policy-making process demonstrated in your example? b) What components...
-
What are the two methods used to translate financial statements and how does the functional currency play a role in determining which method is used?
-
Briefly discuss several data-storage issues that face the modern organization.
-
What is RAID storage technology?
-
How is system performance measurement related to the systems review?
-
Consider the regression model with a single regressor: \(Y_{i}=\beta_{0}+\beta_{1} X_{i}+u_{i}\). Suppose the least squares assumptions in Key Concept 4.3 are satisfied. a. Show that \(X_{i}\) is a...
-
In their study of the effectiveness of cardiac catheterization, McClellan, McNeil, and Newhouse (1994) used as an instrument the difference in distances to cardiac catheterization and regular...
-
In his study of the effect of incarceration on crime rates, suppose Levitt had used the number of lawyers per capita as an instrument. Would this instrument be relevant? Would it be exogenous? Would...

Study smarter with the SolutionInn App



