Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Write Prelab: Purpose, Introduction,...
Fantastic news! We've Found the answer you've been seeking!
Question:

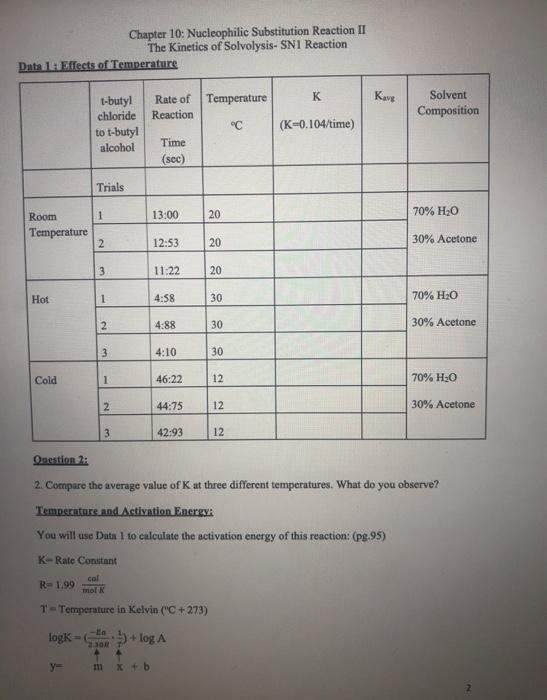

Transcribed Image Text:
Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Write Prelab: Purpose, Introduction, Procedures, and Table of Physical Constants Explanation: In the experiment, we are going to examine the rate of tert-Butyl alcohol+ Cr tert-Butyl chloride + H₂O H₂O in this reaction is a solvent and nucleophile. Sometimes, in an SN1 reaction the solvent acts as the nucleophile. This is called solvolysis. Factors which affect the rate of reaction: A. The concentration of the reactants B. The solvent C. The temperature Concentration of the Reactants: A. The reaction involves two separate steps. First the leaving group departs, and then the nucleophile attacks the carbocation. The first step is the rate-determining step. The rate law for the reaction is as follows: Rate = k [tert-butyl chloride] The rate law doesn't involve water so changing the concentration of the nucleophile has no effect on the rate The rate law does involve [tert-butyl chloride], so increasing the concentration of the substrate increases the rate. Likewise, decreasing the concentration of the substrate decreases the rate. Solvent: B. See explanation for A. Temperature: C. A rise in temperature should accelerate a bond breaking reaction. The rate determining step involves bond breaking. So, we would predict that an increase in temperature should result in production of more tert-butyl and thus increases the rate of the reaction. Question 1: 1. Write the mechanism of the reaction. Data 1: Effects of Temperature Room Temperature Hot Cold 1-butyl chloride to t-butyl alcohol Trials y= 1 2 3. 1 2 3 1 Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction 2 3 Rate of Temperature Reaction Time (sec) m x + b 13:00 12:53 11:22 4:58 4:88 4:10 46:22 44:75 42:93 20 20 T-Temperature in Kelvin ("C +273) logk=+log A 20 30 30 30 12 12 12 °℃ K (K-0.104/time) Kavg Solvent Composition 70% H₂O 30% Acetone 70% H₂O 30% Acetone Question 2: 2. Compare the average value of K at three different temperatures. What do you observe? Temperature and Activation Energy: You will use Data 1 to calculate the activation energy of this reaction: (pg.95) K-Rate Constant col R=1.99 mol K 70% H₂O 30% Acetone Question 3: 3. Plot-log Kang (y axis) vs (x axis) for the three temperatures. Use Excel. Question 4: 4. Why did you have to make graphs? t-butyl chloride to .m-slope 5. Calculate the energy of activation with units. Data 2: Effect of solvent (Read pg. 94) Rate-[tert-Butyl chloride] t-butyl alcohol Trials 1 2 Ea 2.30R 3 Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Time (sec) 9.97 8.03 7.69 Temperature °C 20 20 20 K- 0.104/time Kava Solvent composition 9. Does water participate directly in the reaction? 10. Predict the effect if the solvent were 60% water: 40% acetone. 80% H₂0 20% Acetone Use Data 2 to Answer Questions: 6. Use average to determine K. 7. What is the effect observed from increasing the water from 70% to 80% of the solvent mixture? What about the substrate? 8. What is the function of acetone? I Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Write Prelab: Purpose, Introduction, Procedures, and Table of Physical Constants Explanation: In the experiment, we are going to examine the rate of tert-Butyl alcohol+ Cr tert-Butyl chloride + H₂O H₂O in this reaction is a solvent and nucleophile. Sometimes, in an SN1 reaction the solvent acts as the nucleophile. This is called solvolysis. Factors which affect the rate of reaction: A. The concentration of the reactants B. The solvent C. The temperature Concentration of the Reactants: A. The reaction involves two separate steps. First the leaving group departs, and then the nucleophile attacks the carbocation. The first step is the rate-determining step. The rate law for the reaction is as follows: Rate = k [tert-butyl chloride] The rate law doesn't involve water so changing the concentration of the nucleophile has no effect on the rate The rate law does involve [tert-butyl chloride], so increasing the concentration of the substrate increases the rate. Likewise, decreasing the concentration of the substrate decreases the rate. Solvent: B. See explanation for A. Temperature: C. A rise in temperature should accelerate a bond breaking reaction. The rate determining step involves bond breaking. So, we would predict that an increase in temperature should result in production of more tert-butyl and thus increases the rate of the reaction. Question 1: 1. Write the mechanism of the reaction. Data 1: Effects of Temperature Room Temperature Hot Cold 1-butyl chloride to t-butyl alcohol Trials y= 1 2 3. 1 2 3 1 Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction 2 3 Rate of Temperature Reaction Time (sec) m x + b 13:00 12:53 11:22 4:58 4:88 4:10 46:22 44:75 42:93 20 20 T-Temperature in Kelvin ("C +273) logk=+log A 20 30 30 30 12 12 12 °℃ K (K-0.104/time) Kavg Solvent Composition 70% H₂O 30% Acetone 70% H₂O 30% Acetone Question 2: 2. Compare the average value of K at three different temperatures. What do you observe? Temperature and Activation Energy: You will use Data 1 to calculate the activation energy of this reaction: (pg.95) K-Rate Constant col R=1.99 mol K 70% H₂O 30% Acetone Question 3: 3. Plot-log Kang (y axis) vs (x axis) for the three temperatures. Use Excel. Question 4: 4. Why did you have to make graphs? t-butyl chloride to .m-slope 5. Calculate the energy of activation with units. Data 2: Effect of solvent (Read pg. 94) Rate-[tert-Butyl chloride] t-butyl alcohol Trials 1 2 Ea 2.30R 3 Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Time (sec) 9.97 8.03 7.69 Temperature °C 20 20 20 K- 0.104/time Kava Solvent composition 9. Does water participate directly in the reaction? 10. Predict the effect if the solvent were 60% water: 40% acetone. 80% H₂0 20% Acetone Use Data 2 to Answer Questions: 6. Use average to determine K. 7. What is the effect observed from increasing the water from 70% to 80% of the solvent mixture? What about the substrate? 8. What is the function of acetone? I
Expert Answer:
Answer rating: 100% (QA)
Question we observed that as temperature is higher than rate of react... View the full answer

Related Book For 

Human Resource Management
ISBN: 978-0078029127
12th edition
Authors: John Ivancevich, Robert Konopaske
Posted Date:
Students also viewed these management leadership questions
-
What personal qualities do you think are necessary for an employee to be successful at a company such as Zappos? How would you select for those qualities? Explain.
-
What social media platforms do you think are most critical for Lyon College to engage?
-
What services do you think are most important to todays increasingly diverse workforce? Why?
-
Products are 1. Lip gloss or lipstick with SPF 2. Custom facial sheets you can make online or in store pickup and have it send to your home based on your needs 3. A foundation sunscreen Price Provide...
-
Nationally, 38% of fourth-graders cannot read an age-appropriate book. The following data show the number of children, by age, identified as learning disabled under special education. Most of these...
-
In Problem 52, part (b), solve assuming \(k_{1}=k_{2}=\) \(k_{3}=k_{4}=k\) and \(m_{1}=m_{3}=m\) and \(m_{2}=m(1+\epsilon)\). Use the same parameter values. Discuss. Problem 52: (b) For the previous...
-
The following MINITAB output presents a 95% confidence interval for the mean cholesterol levels for men aged 50 years, and a 95% prediction interval for an individual man aged 50. The units of...
-
A spherical water drop is suspended from a fine thread in still, dry air. Show: (a) That the Sherwood number for mass transfer from the surface of the drop into the surroundings has a value of 2 if...
-
Lydex Company s financial statements for the last two years are as follows:Lydex CompanyComparative Balance Sheet This YearLast YearAssets Current assets: Cash$ 8 6 0 , 0 0 0 $ 1 , 1 0 0 , 0 0 0...
-
You are considering a purchase of a 4-plex, which is located in a desirable neighborhood. The cost of the property is $500,000. Effective rents are expected to average $1500 per month. Every resident...
-
Define random variable X as the annual rainfall in Chicago, which is a valuable metric for the local agriculture industry. From climatologically data, we can assume X has a normal distribution with...
-
Helium atomic number is 2, atomic weight is 4.0026amu. How many protons and electrons are in a helium atom? Mass of proton is 1.0007amu, mass of neutron is 1.0008amu. If there are only 2, isotopes of...
-
Compute the cost assigned to ending inventory using fifo, lifo, weighted average, and specific identification. for specific identifications, units sold include 4 5 units from beginning inventory, 1 7...
-
A 2.0-kg object is attached to a 1.5-m long string and swung in a vertical circle at a constant speed of 12 m/s. a. What is the tension in the string when the object is at the bottom of its path? b....
-
Discuss the implications of advanced constitutive models, like the Cam-Clay and Mohr-Coulomb models, in understanding the stress-strain behavior of soils under cyclic loading, and how this...
-
In the figure, the particles have charges q1 = -92=610 nC and 93 94 92 nC, and distance a = 4.9 cm. What are the (a) x and (b) y components of the net electrostatic force on particle 3? Y 3 12 (a)...
-
3. Conceptual Connection: Job 73 used 20 machine hours from Department A and 50 machine hours from Department B. Job 74 used 50 machine hours from Department A and 20 machine hours from Department B....
-
All of the following assets can be depreciated, except: (a) A bulldozer (b) A copper mine (c) A surgical robot (d) A conveyor belt
-
What defines career success to you?
-
Why are some people satisfied with what is identified as a midcareer plateau while others at the same point experience career burnout?
-
Describe the current trends in union membership and union organizing.
-
Why are provisions a problem?
-
Who are the main users of financial accounting information, and what are their information needs?
-
Why do we not have pure historic cost accounting?

Study smarter with the SolutionInn App


