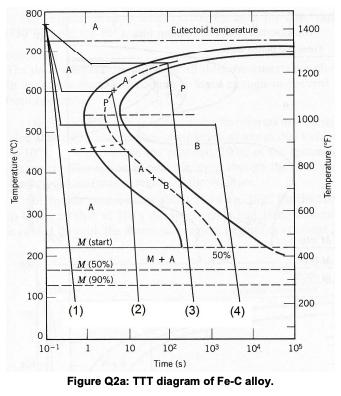
Based on the following Time-Temperature transformation diagram for an iron-carbon alloy of eutectoid composition (Figure Q2a),...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Based on the following Time-Temperature transformation diagram for an iron-carbon alloy of eutectoid composition (Figure Q2a), determine the final microstructure of a small specimen that has been subjected to the following time-temperature treatments (1), (2), (3), and (4), as shown in the Figure Q2a. In each case assume that the specimen temperature begins at around 760 oC, and that it has been held at this temperature long enough to have achieved a complete and homogeneous Austenitic structure. Q2) Two previously un-deformed cylindrical specimens of brass are to be cold worked by reducing their cross-sectional areas (while maintaining their circular cross- sections). For the first specimen, the initial and deformed radii are 16 and 11 mm, respectively. The second specimen, with an initial radius of 12 mm, must have the same deformed hardness as the first specimen. Calculate the second specimen's radius after deformation. Q3)The alloy is cooled down to 720 °C, please answer the following questions: How many phases are present and what are their names? For the phases identified above, what are their compositions for each phase? Sketch the micrograph, taking into account the features of the phase diagram. What is the weight fraction of each phase? What is the weight fraction for pearlite in the alloy? Temperature (°C) 800 700 600 500 400 300 200 100 0 10-1 A M (start) A M (50%) M (90%) (1) 1 P-X (2) 10 Eutectoid temperature M + A P B (3) 50% 10² Time (s) Figure Q2a: TTT diagram of Fe-C alloy. (4) 10³ 104 1400 1200 1000 800 600 400 200 10⁰ Temperature (°F) Based on the following Time-Temperature transformation diagram for an iron-carbon alloy of eutectoid composition (Figure Q2a), determine the final microstructure of a small specimen that has been subjected to the following time-temperature treatments (1), (2), (3), and (4), as shown in the Figure Q2a. In each case assume that the specimen temperature begins at around 760 oC, and that it has been held at this temperature long enough to have achieved a complete and homogeneous Austenitic structure. Q2) Two previously un-deformed cylindrical specimens of brass are to be cold worked by reducing their cross-sectional areas (while maintaining their circular cross- sections). For the first specimen, the initial and deformed radii are 16 and 11 mm, respectively. The second specimen, with an initial radius of 12 mm, must have the same deformed hardness as the first specimen. Calculate the second specimen's radius after deformation. Q3)The alloy is cooled down to 720 °C, please answer the following questions: How many phases are present and what are their names? For the phases identified above, what are their compositions for each phase? Sketch the micrograph, taking into account the features of the phase diagram. What is the weight fraction of each phase? What is the weight fraction for pearlite in the alloy? Temperature (°C) 800 700 600 500 400 300 200 100 0 10-1 A M (start) A M (50%) M (90%) (1) 1 P-X (2) 10 Eutectoid temperature M + A P B (3) 50% 10² Time (s) Figure Q2a: TTT diagram of Fe-C alloy. (4) 10³ 104 1400 1200 1000 800 600 400 200 10⁰ Temperature (°F)
Expert Answer:
Answer rating: 100% (QA)
1 Given Initial radius of 1st specimen 16 mm Deformed radius of ... View the full answer

Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-0470419977
8th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these accounting questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
John is the sole owner of 'Dude Mug Shop'. The business is registered for GST. He has provided you with the following information in relation to the 2021 FY budget: Estimated sales units for the...
-
Two moles of an ideal monatomic gas go through the cycle abc. For the complete cycle, 800 J of heat flows out of the gas. Process ab is at constant pressure, and process bc is at constant volume....
-
In 2011, the industries with the most complaints to the Better Business Bureau were banks, cable and satellite television companies, collection agencies, cellular phone providers, and new car...
-
Search the Internet and find those countries in which financial markets are governed by Islamic law. Which countries have the strictest interpretation of the Qur'an, and which countries have a more...
-
Assuming that the necessary assumptions are met, construct a 95% confidence interval for \(ho\) when (a) \(r=0.72\) and \(n=19\); (b) \(r=0.35\) and \(n=25\); (c) \(r=0.57\) and \(n=40\).
-
Read the PCAOB enforcement case against Christopher E. Anderson (PCAOB Release No. 105-2008-003, available at www.pcaob.org). Among the PCAOB's findings is that the audit partner, Anderson, changed...
-
Suppose Carol's stock price is currently $20 and does not currently pay dividends. In the next 6 months it will either fall to $10 or rise to $40. What is the current value of a European 6-month call...
-
Absorption Costing Versus Variable Costing. Prime, Inc., produces tricycles. The company has no finished goods inventory at the beginning of year 1. The following information pertains to Prime, Inc....
-
Geza ordered custom-made kitchen cabinets from a reputable manufacturer located in Italy. The contract stated that the completed cabinets would be delivered to Geza's home by a date set by the...
-
Read the article "Needle and syringe programmes are highly cost-effective at preventing hepatitis C transmission" and answer the questions below. At what point in the research might the researchers...
-
Blossom enters into an agreement with Crane Inc. to lease a car on December 31, 2024. The following information relates to this agreement. 1. The term of the non-cancelable lease is 3 years with no...
-
If YOU were to devise a personal strategy for how to behave in challenging interpersonal situations, outline the steps to this strategy. In addition, indicate to what degree would this strategy use...
-
Last year, Harold Company produced 2,000 units and sold 1,000 units. The company had no beginning inventory. Harold incurred the following costs: Direct materials per unit Direct labor per unit $30...
-
Develop a short service quality improvement plan for the police department as a set of instructions from improving its service quality.
-
On June 8, Cato Ltd. was incorporated and issued 26,400 common shares for $264,000. On August 19, an additional 6,600 shares were issued for $79,200. On November 2, the company paid $25,344 to...
-
Consider the reaction of acetic acid in water CH 3 CO 2 H(aq) + H 2 O(l) CH3CO 22 (aq) + H 3 O + (aq) where Ka 5 1.8 3 1025. a. Which two bases are competing for the proton? b. Which is the stronger...
-
(a) Is it possible to grind up and reuse phenol-formaldehyde? Why or why not? (b) Is it possible to grind up and reuse polypropylene? Why or why not?
-
The preexponential and activation energy for the diffusion of iron in cobalt are 1.1 10-5 m2/s and 253,300 J/mol, respectively. At what temperature will the diffusion coefficient have a value of 2.1...
-
How would you expect a decrease in the austenite grain size to affect the harden ability of a steel alloy? Why?
-
If two countries had identical term structures of interest rates, what is the expected future exchange rate change between the two currencies?
-
Pick three developing countries and create a country risk index for them. Rank them ordinally in terms of factors that you can observe (exports, GDP growth, reserves, etc.) by looking at...
-
Give examples of policies that a country could implement to reduce its absorption.

Study smarter with the SolutionInn App


