Bond enthalpy is the energy required to break a mole of a certain type of bond....
Fantastic news! We've Found the answer you've been seeking!
Question:
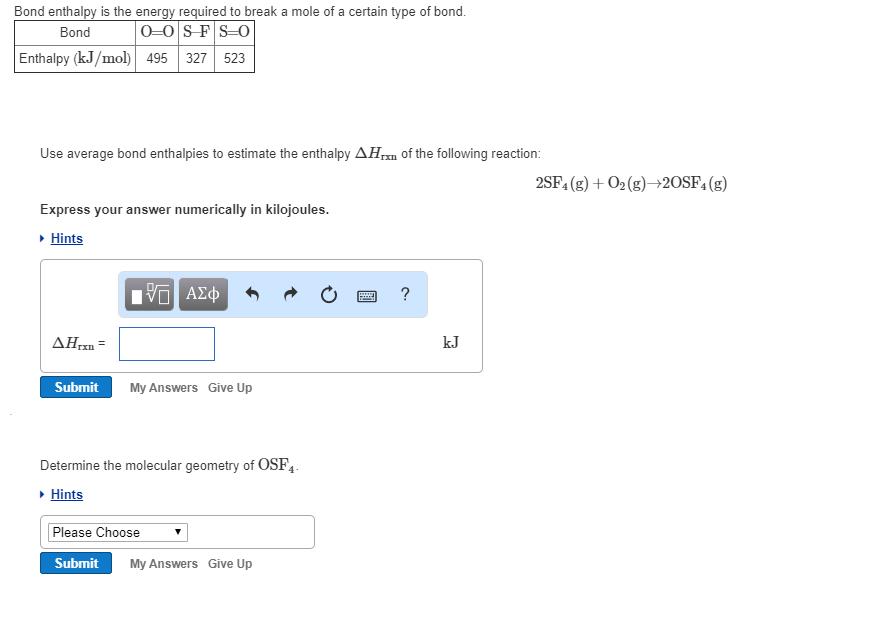
Transcribed Image Text:
Bond enthalpy is the energy required to break a mole of a certain type of bond. 0-0 SFS-0 Bond Enthalpy (kJ/mol) 495 327 523 Use average bond enthalpies to estimate the enthalpy AH of the following reaction: Ixn Express your answer numerically in kilojoules. ▸ Hints VE ΑΣΦ AHrxn= kJ Submit My Answers Give Up Determine the molecular geometry of OSF4. ▸ Hints Please Choose Submit My Answers Give Up 2SF4 (g) + O₂(g) →2OSF4 (g) Bond enthalpy is the energy required to break a mole of a certain type of bond. 0-0 SFS-0 Bond Enthalpy (kJ/mol) 495 327 523 Use average bond enthalpies to estimate the enthalpy AH of the following reaction: Ixn Express your answer numerically in kilojoules. ▸ Hints VE ΑΣΦ AHrxn= kJ Submit My Answers Give Up Determine the molecular geometry of OSF4. ▸ Hints Please Choose Submit My Answers Give Up 2SF4 (g) + O₂(g) →2OSF4 (g)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The dissociation energy of a molecule is the energy required to break the molecule apart into its separate atoms. The dissociation energy for the cyanogen molecule is 1.22 Ã 10-18 J. Suppose...
-
The work function of an element is the energy required to remove an electron from the surface of the solid. The work function for lithium is 279.7 kJ/ mol (that is, it takes 279.7 kJ of energy to...
-
The energy required to increase the speed of a certain car from 18 m/s to 24 m/s is 190 kJ. (a) What is the mass of the car? (b) At what speed is the car's kinetic energy equal to 190 kJ?
-
Calculate the anion gap for a 58 year old insulin dependent diabetic woman who is admitted to the emergency room in a comatose state Na-135mmol/L K= 3.5 mmol/L CI-102 mmol/L HCO3-= 15mmol/L. Include...
-
England Corporation is issuing $200,000 of 8%, 5-year bonds when potential bond investors want a return of 10%. Interest is payable semiannually. Instructions Compute the market price (present value)...
-
Although HCl and H2SO4 have very different properties as pure substances, their aqueous solutions possess many common properties. List some general properties of these solutions, and explain their...
-
A contingency table containing observed values has three rows and four columns. The number of degrees of freedom for the chi-square statistic is 7. In Exercises 68, determine whether the statement is...
-
Cole Petroleum has spent $206,000 to refine 63,000 gallons of petroleum distillate, which can be sold for $6.30 a gallon. Alternatively, Cole can process the distillate further and produce 53,000...
-
A donation, on 27 March 2017, between Rose and Andre, her "ninong". Here, Rose donated a parcel of land with an area of 5,000 square meters to Andre. When you were talking, Rose told you that she...
-
Identify the main operator in the following propositions. 1. (A M ) (C E ) 2. (G P) (H W) 3. [P (S K)] 4. (K O) (R B) 5. (M B) [E (C I)] 6. [(P R) (E F)] 7. [(S L) M] (C N)...
-
The data shown below were obtained on a highway. Use regression analysis to fit these data to the Greenshields model and determine (a) the mean free speed, (b) the jam density, (c) the capacity, and...
-
What are some situations other than immediate financial distress that lead firms to file for bankruptcy?
-
What happened to the pre-bankruptcy stockholders and the claims of creditors?
-
What is the annual report, and what two types of information does it present?
-
The name under which a fi rm does its business. a. goodwill b. trade na me c. trade s ecret d. brand na me e. trademark
-
In general, how much do unsecured creditors receive from a liquidation? How much do stockholders receive?
-
Evaluate 50 S T u (ln (+)-1) du
-
Calculate I, , and a for a 0.0175 m solution of Na 3 PO 4 at 298 K. Assume complete dissociation. How confident are you that your calculated results will agree with experimental results?
-
Arrange the following species in order of increasing stability: Li2, Li2+, Li2-2. Justify your choice with a molecular orbital energy level diagram?
-
The H- ion and the He atom have two 1s electrons each. Which of the two species is larger? Explain.
-
Calculate the concentration (in molarity) of a NaOH solution if 25.0mL of the solution are needed to neutralize 17.4mL of a 0.312 M HCl solution.
-
Discuss the similarities and differences betw1een Maslows hierarchy of needs theory and E.R.G. theory. What insights do you gain from each theory?
-
Discuss the different ways of expressing the need for power as described in McClellands achievement motivation theory. Which of the two types of power do you respond to best? Why?
-
Which parts of Herzbergs motivatorhygiene theory made useful contributions to understanding motivation in organizations?

Study smarter with the SolutionInn App



