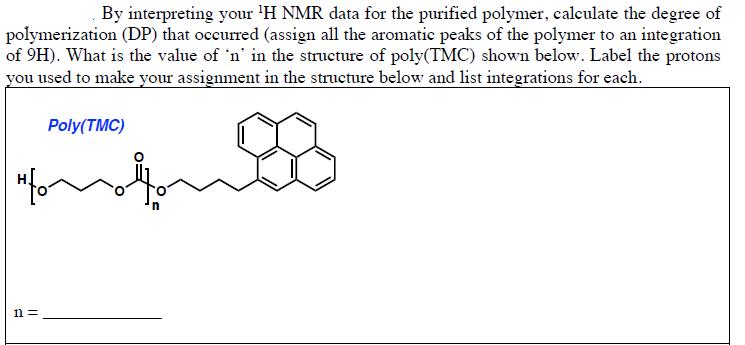
By interpreting your 'H NMR data for the purified polymer, caleulate the degree of polymerization (DP)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
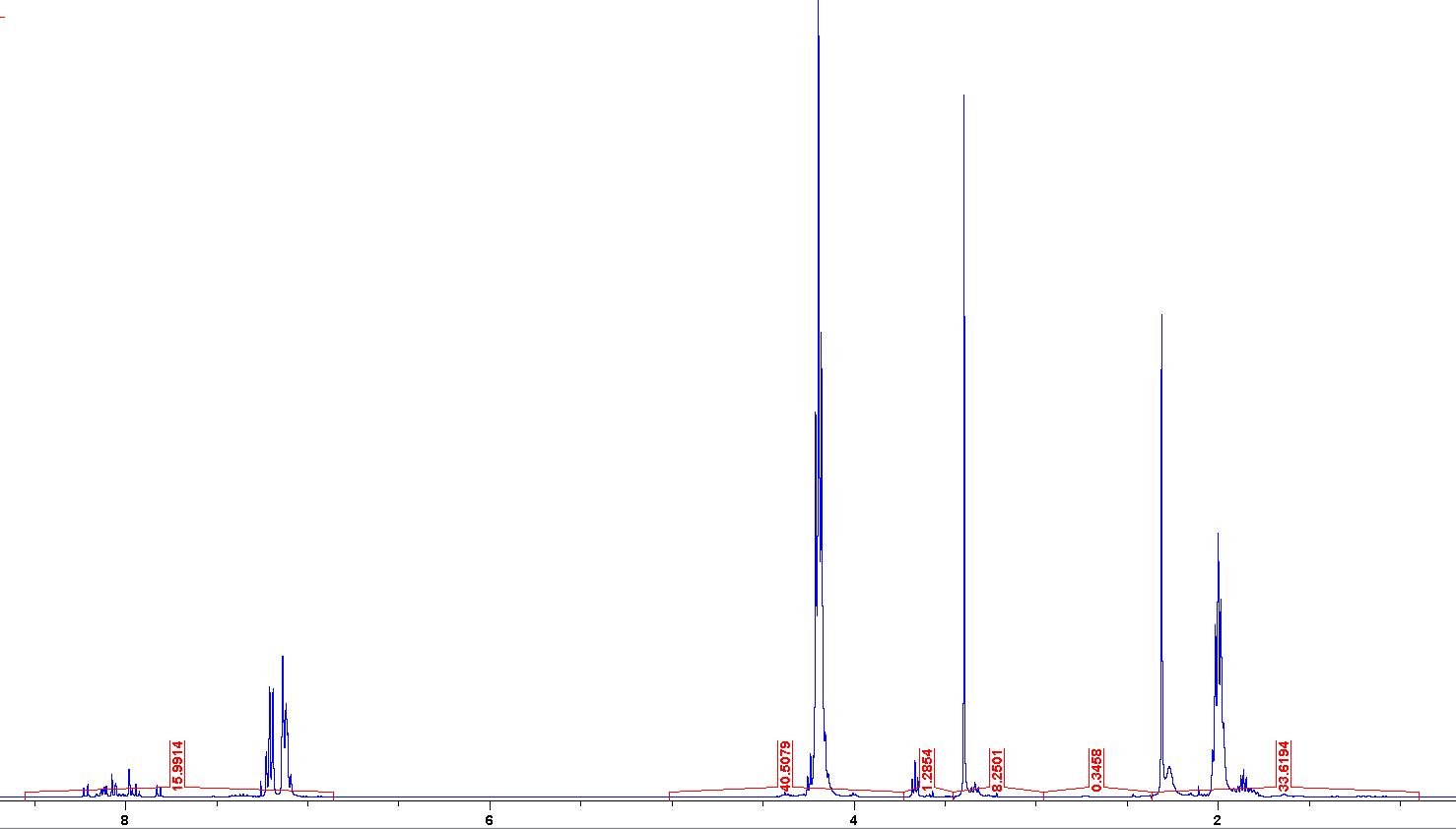
By interpreting your 'H NMR data for the purified polymer, caleulate the degree of polymerization (DP) that occurred (assign all the aromatic peaks of the polymer to an integration of 9H). What is the value of 'n' in the structure of poly(TMC) shown below. Label the protons you used to make your assignment in the structure below and list integrations for each. Poly(TMC) "fon n = 15.9914 1.2854 8.2501 0.3458 33.6194 By interpreting your 'H NMR data for the purified polymer, caleulate the degree of polymerization (DP) that occurred (assign all the aromatic peaks of the polymer to an integration of 9H). What is the value of 'n' in the structure of poly(TMC) shown below. Label the protons you used to make your assignment in the structure below and list integrations for each. Poly(TMC) "fon n = 15.9914 1.2854 8.2501 0.3458 33.6194
Expert Answer:
Answer rating: 100% (QA)
By interpreting your H NMR data for the purified polymer calculate the degree of p... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
The 1H NMR data for the two anomers included very comparable peaks in the d 2.0-5.6 region but differed in that, as their highest peaks, anomer V had a doublet at d 5.8 (1H, J = 12 Hz) while anomer...
-
a. What is the value of n in Hckel's rule when a compound has nine pairs of electrons? b. Is such a compound aromatic?
-
The degree of polymerization of poly tetra fluoro ethylene (see Table 16-3) is 7500. If all of the polymer chains are the same length, calculate (a) The molecular weight of the chains, and (b) The...
-
Courts are more willing to find misrepresentation if the defendant has a fiduciary relationship with the plaintiff than if a transaction occurs at arms length between the parties. True False
-
What recent developments have caused supply chain management to become increasingly important to international companies?
-
The Drosselmeier Corporation, located in Munich, makes Christmas nutcrackers and has an annual plant capacity of 2,400 product units. Suppose its predicted operating results (in euros) for the year...
-
Review your answers to the previous two questions. Comment on the persuasiveness of evidence from each test. Explain any factors that would affect your assessment. Fellowes and Associates Chartered...
-
Lockovich borrowed money from a bank to purchase a motorboat. The bank took a security interest in it but never filed a financing statement. A subsequent default on the loan occurred, and the debtor...
-
Ex6: Identify and correct the error(s) in each of the following: a) class Bag extends Product { ... } Bag b1 = new Product(); b) class Vehicle { abstract private getVIN(); ... }
-
The worksheet used to produce Figure 23.2 is on the authors web site. How does the loss distribution change when the loss severity has a beta distribution with upper bound of 5, lower bound of zero,...
-
From the following information find out the amount of profit earned during the year: Fixed cost=$600,000 Variable cost=$12 per unit Selling price=$18 per unit Output level=170,000 units
-
A car is \(12 \mathrm{~m}\) from the bottom of a ramp that is \(8.0 \mathrm{~m}\) long at its base and \(6.0 \mathrm{~m}\) high (Figure P3.81). The car moves from rest toward the ramp with an...
-
Modified quotas are given in Problems 7-14. Round your answers to two decimal places. a. Find the lower and upper quotas. b. Find the arithmetic mean of the lower and upper quotas. c. Find the...
-
Consider the graph of \(y=x^{2}\) bounded by the \(x\)-axis and the line \(x=2\). Approximate the area under the curve by using rectangles and right endpoints as described in Problems 47-52. Use four...
-
In 2015, Susans sole proprietorship earns $300,000 of self-employment net income (after the deduction for one-half of self-employment tax). a. Calculate the maximum amount Susan can deduct for...
-
Consider the following classical economy: \[ \begin{array}{ll} A D & Y=400+50 \mathrm{M} / \mathrm{P} \\ A S & Y=\bar{Y}=1000 . \end{array} \] This economy produces only wine, its output is measured...
-
Part 1: Informative Speech Plan Include a list of the ten topics that you had initially considered while you were generating your initial list of brainstormed topics. Explain your process of...
-
United Business Forms capital structure is as follows: Debt ............................................ 35% Preferred stock ........................... 15 Common equity .......................... 50...
-
(a) What factor explains the observation that tertiary alcohols react with HX faster than secondary alcohols? (b) What factor explains the observation that methanol reacts with HX faster than a...
-
Arrange the following in order of increasing basicity: (a) (b) (c)
-
Compound Y (C8H4O3) dissolves slowly when warmed with aqueous sodium bicarbonate. The IR spectrum of Y has strong peaks at 1779 and at 1854 cm-1. The broadband proton-decoupled 13C spectrum of Y...
-
Nestls financial statements are presented in Appendix B. Financial statements of Delfi Limited are presented in Appendix C. Instructions Refer to the financial statements and answer the following...
-
Writing Financial statement information about four different companies is as follows. Instructions a. Determine the missing amounts. (Hint: For example, to solve for (a), Assets Liabilities = Equity...
-
The financial statements of TSMC are presented in Appendix A. The complete annual report, including the notes to the financial statements, is available at the companys website. Instructions Refer to...

Study smarter with the SolutionInn App


