c. Challenge Problem Suppose a total of 15 ounces of medicine is added to the original...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
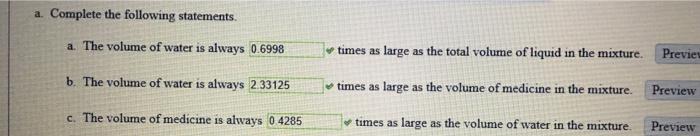
c. Challenge Problem Suppose a total of 15 ounces of medicine is added to the original mixture (so that the total volume is now 25 ounces with 18 ounces of medicine and 7 ounces of water). How much water must now be added so that the mixture has the same proportion of medicine and water as the original mixture? ounces Preview Try again. If there is 18 ounces of medicine in the mixture, how many ounces of water should there be in the mixture (use the ratios you computed earlier in this example)? How many ounces of water must you add to the 7 ounces of water to obtain this amount of water? a. Complete the following statements. a. The volume of water is always 0.6998 b. The volume of water is always 2.33125 c. The volume of medicine is always 0.4285 times as large as the total volume of liquid in the mixture. Preview times as large as the volume of medicine in the mixture. Preview times as large as the volume of water in the mixture. Preview c. Challenge Problem Suppose a total of 15 ounces of medicine is added to the original mixture (so that the total volume is now 25 ounces with 18 ounces of medicine and 7 ounces of water). How much water must now be added so that the mixture has the same proportion of medicine and water as the original mixture? ounces Preview Try again. If there is 18 ounces of medicine in the mixture, how many ounces of water should there be in the mixture (use the ratios you computed earlier in this example)? How many ounces of water must you add to the 7 ounces of water to obtain this amount of water? a. Complete the following statements. a. The volume of water is always 0.6998 b. The volume of water is always 2.33125 c. The volume of medicine is always 0.4285 times as large as the total volume of liquid in the mixture. Preview times as large as the volume of medicine in the mixture. Preview times as large as the volume of water in the mixture. Preview
Expert Answer:
Answer rating: 100% (QA)
a The load complex power are V 200 Zi 08j56 V 200 S Z 8 j16 S3 50003687 4000j3000 VA Therefor... View the full answer

Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Posted Date:
Students also viewed these mathematics questions
-
If circulation K is added to the cylinder flow in Prob. 8.43, (a) for what value of K will the flow begin to cavitate at the surface? (b) Where on the surface will cavitation begin? (c) For this...
-
If a 2% charge is added to the annual premium of $ 1,021.80 when payments are made semiannually, how much would semiannual payments be?
-
In Prob. 5-173, presume that air is added to the tank in such a way that the temperature and pressure of the air in the tank remain constant. Determine the final mass of the air in the ballast tank...
-
Every real number is either a/an number or a/an_______ number.
-
Tom Sharkey is the owner of Sharkey Chevy, Buick, GMC. At the start of the year, Tom instituted a customer opinion program to find ways to improve service. The day after the service is performed,...
-
Given the stresses in psi: xx = 1,500 yy = 2,000 zz = 3,500 xy = 600 yz = 300 zx = 500 Where x = east, y = north, z = up, compression is positive, find: the secondary principal stresses in the...
-
Suppose that in September 2013 a company takes a long position in a contract on May 2014 crude oil futures. It closes out its position in March 2014. The futures price (per barrel) is \($88.30\) when...
-
Adria Lopez, owner of Success Systems, realizes that she needs to begin accounting for bad debts expense. Assume that Success Systems has total revenues of $ 43,853 during the first three months of...
-
Consider the acceleration of a good sports car in real life . (1) Pick a certain car model, which could be the one you are driving, or the one you wish you were --- and search online for its...
-
In Section 1.4, the analytics study team was tasked with making a recommendation for the best level of advertising for the VRX2000 during the upcoming first quarter (Q1). They used historical data...
-
Evaluate the statement by Thomas Friedman that the world is flat. Friedman analyzes globalization in the early21st century. The title is a metaphor for viewing the world as a level playing field in...
-
Please write a letter of recommendation for position of employment in an organization. And make sure it is maximum of 3paragraph with the template of introduction, body and conclusion. (Please do...
-
Suppose Jeff Bezos deposits $1 million in currency in his checking account on the Moon. Calculate the change in the money supply on the Moon that follows this deposit.
-
The current cost of graduate school tuition is $12,000 per year. The cost of tuition is rising at 5.00% per year. You plan to attend graduate school for 2 years starting 2 years from now. How much do...
-
What are the prominent distinctions and commonalities in global leadership styles, as delineated by pertinent constructs? Furthermore, do leaders originating from particular cultural backgrounds...
-
Steven and Sally Smith are spouse and they have two children, Jamila aged 15 and Nic aged 12, both of whom will be dependent until age 18. Steven and Sally are not sure what would be the cost of...
-
Selected amounts from the accounting records of White Birch Technology follow. (Click the icon to view the financial data) Required 1. Prepare White Birch's income statement for the year ended...
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
a What trend in volatility is seen going down Group 17? b. Using Table 12.1, what is the state of each halogen at 20C? c. What is the trend in the atomic radii of the halogens? Explain this trend. d....
-
The table shows the type of bonding in a number of elements and compounds. a. Draw a labelled diagram to show metallic bonding. b. Explain why magnesium chloride has a high melting point but bromine...
-
a. Write a balanced equation for the reaction that takes place when propanal is warmed with an aqueous alkaline solution of sodium tetrahydridoborate, using the symbol [H] to represent a hydrogen...
-
The figure below illustrates a thin plate of thickness \(t\). An approximate displacement field, which accounts for displacements due to the weight of the plate, is given by a. Determine the...
-
For a plane stress problem, the strain components in the xyplane at a point \(P\) are computed as: a. Compute the state of stress at this point if Young's modulus \(E=2 \times 10^{11} \mathrm{~Pa}\)...
-
The stress matrix at a particular point in a body is Determine the corresponding strain if \(E=20 \times 10^{10} \mathrm{~Pa}\) and \(v=0.3\). -2 1-3 [0] = 104 -345 107 Pa.

Study smarter with the SolutionInn App


