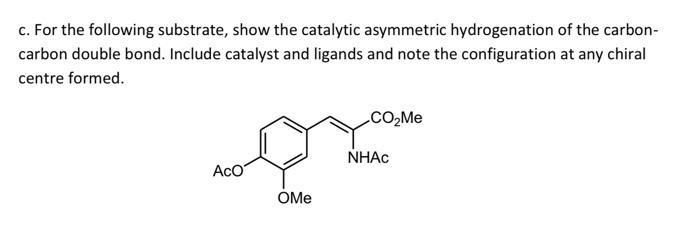
c. For the following substrate, show the catalytic asymmetric hydrogenation of the carbon- carbon double bond....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
c. For the following substrate, show the catalytic asymmetric hydrogenation of the carbon- carbon double bond. Include catalyst and ligands and note the configuration at any chiral centre formed. .CO2M. NHAC AcO OMe c. For the following substrate, show the catalytic asymmetric hydrogenation of the carbon- carbon double bond. Include catalyst and ligands and note the configuration at any chiral centre formed. .CO2M. NHAC AcO OMe
Expert Answer:
Answer rating: 100% (QA)
Solution Required reaction mechanism is as follows Asy... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the standard free-energy change at 25C for the following reaction. 3Cu(s) + 2NO3 (aq) + 8H+(aq) 3Cu2+(aq) + 2NO(g) + 4H2O(l)
-
Calculate the standard free-energy change at 25C for the following reaction. 4Al(s) + 3O2(g) + 12H + (aq) 4Al3 + (aq) + 6H2O(l) Use standard electrode potentials.
-
For each structure, 1. Star (*) any asymmetric carbon atoms. 2. Label each asymmetric carbon as (R) or (S). 3. Draw any internal mirror planes of symmetry. 4. Label the structure as chiral or...
-
Example Problem for Declining Balance Depreciation Methods Historical cost of equipment A: $120,000, purchased on January 1, 2014. Estimated salvage value: $20,000; Estimated useful life: 5 years....
-
Following up Problem 16.22, even though we take riskiness into account, there still is difficulty with NPV as a decision criterion. Sup-pose that you are facing the two risky projects shown in Figure...
-
Following are several independent accounting situations: 1. Interest of $1,500 on a loan outstanding during 2012 is due on January 1, 2013. 2. Received $100,000 in 2012 for a service project that is...
-
The Eaton Company was incorporated July 10, 19X0, with an authorized capital as follows: 1. Common stock, Class A, 20,000 shares, par value \(\$ 25\) per share. 2. Common stock, Class \(B, 100,000\)...
-
At the beginning of 2016, Better Corp.s accounting records had the following general ledger accounts and balances. Better Corp. completed the following transactions during 2016: 1. Purchased land for...
-
Weight Watchers really wants to let you know that it's not just a diet company -- so much so that it's changing its name to WW. The company announced Monday that the new WW (WTW) name reflects its...
-
The three forces are applied to the bracket. Determine the range of values for the magnitude of force P so that the resultant of the three forces does not exceed 2400 N. 3000 N 90%- Prob. 2-38 800 N...
-
In order to justify the cost of using a wire transfer instead of an EDT (electronic depository transfer), your company must transfer a minimum of $200,000. The EDT has a one business day...
-
John Deere Inc is evaluating the revenues expected to be generated by a new product. They will evaluate it as if the product will generate $ 2 3 million per year in revenues for the first 1 0 years (...
-
The records of Bonita's Boutique report the following data for the month of April. Sales revenue $96,600 Purchases (at cost) $47,900 Sales returns 2,000 Purchases (at sales price) 91,000 Markups...
-
Trial Balance When should this be done by? End of the month Who should complete this report? A) Explain how this figure is achieved B) What records are used? What does it represent? Cashbook...
-
Question 3 Zenith Ltd is a company located in Midrand that manufactures and distributes electronic products and suppliers. The following information is available for the year ended 28 February 2023...
-
Ayayai Company lost most of its inventory in a fire in December just before the year-end physical inventory was taken. The corporation's books disclosed the following. Beginning inventory $177,000...
-
Problem 4 (6 points) Suppose g(x) is a monic polynomial of even degree d, that is g(x) = xd +bd-1xd1 + +6x+bo ...
-
A copper wire (density = 8.96 g/cm 3 ) has a diameter of 0.25 mm. If a sample of this copper wire has a mass of 22 g, how long is the wire?
-
For each pair of compounds, predict the one with a higher boiling point. Which compounds have zero dipole moments? (a) cis-1,2-dichloroethene or cis-1,2-dibromoethene (b) cis- or...
-
(a) Rank ethanol, methylamine, and acetic acid in decreasing order of acidity. (b) Rank ethanol, methylamine (pKb 3.36), and ethoxide ion (CH3CH2O-) in decreasing order of basicity. In each case,...
-
An inexperienced graduate student treated dec-5-ene with borane in THF, placed the flask in a refrigerator, and left for a party. When he returned from the party, he discovered that the refrigerator...
-
Trust Management System: A university would like to develop a system for students to evaluate their peers performance in some group projects. Students ratings in various categories will affect a...
-
Building trust in potential students to join music classes by a music teacher a. Explain the above scenario. b. Draw a class diagram for it. c. Document a detailed and significant use case. d. Create...
-
Building trust in parents by hostel authorities to state that hostel is safe for students to live a. Explain the above scenario. b. Draw a class diagram for it. c. Document a detailed and significant...

Study smarter with the SolutionInn App


