2. A 25.0 g sample of nitrogen gas, N, has a volume of 50.0 L and...
Fantastic news! We've Found the answer you've been seeking!
Question:



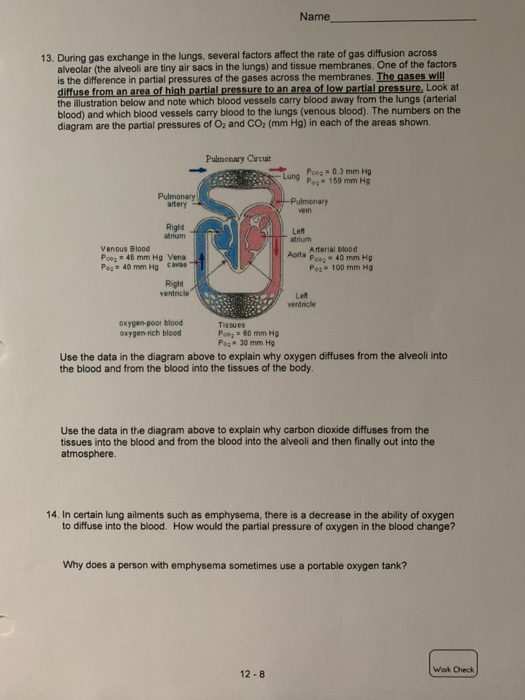
Transcribed Image Text:
2. A 25.0 g sample of nitrogen gas, N₂, has a volume of 50.0 L and a pressure of 635 mm Hg. What is the temperature of the gas? Work Check 3. How many molecules of gas will fill a 30.0 gallon trash bag at 25 °C and 760. mm Hg? (1 L = 1.06 qts; 1 gal = 4 qts) 4. Aerosol containers can be dangerous if they are heated because they can explode. Suppose a container of hair spray with a pressure of 4.0 atm at a room temperature of 25 °C is thrown into a fire. a) If the temperature of the gas inside the aerosol can reaches 402 °C, what will the pressure be? b) The aerosol container may explode if the pressure inside exceeds 8.0 atm. Would you expect it to explode? 12-5 Wok Check Name 5. A volume of 500. mL of air on a cold winter day at 5 °C is inhaled into the lungs. The body temperature is 37 °C. What will be the new volume of the air in the lungs? Assume the pressure remains constant at 760 mm Hg. 6. The volume of air in a person's lungs is 615 mL at a pressure of 760. mm Hg. Inhalation occurs as the pressure in the lungs drops to 752 mm Hg. To what volume did the lungs expand? 7. A sample of gas containing 1.50 moles of neon has a volume of 8.50 L. What will be the volume of the sample of gas if a leak allows one-half of the neon atoms to escape? 8. A sample containing 4.80 g of oxygen gas, O₂, has a volume of 15.0 L. Oxygen gas is released until the volume is 10.0 L. How many moles of oxygen are removed? 12-6 C Name 9. What volume will 3.5g of CO occupy at 17 °C and 1875 torr? 10. A typical air sample in the lungs contains oxygen at 100 mm Hg, nitrogen at 573 mm Hg, carbon dioxide at 40 mm Hg, and water vapor at 47 mm Hg. Why are these pressures called partial pressures? 11. An anesthetic consists of a mixture of cyclopropane gas, C₂H₁, and oxygen gas, O₂. If the mixture has a total pressure of 825 torr, and the partial pressure of the cyclopropane gas is 73 torr, what is the partial pressure of the oxygen in the anesthetic? 12. If 74.5 % of exhaled air is nitrogen, what is the partial pressure of nitrogen in exhaled air. Assume that the total pressure of exhaled air is 760. mm Hg. 12-7 Work Check 13. During gas exchange in the lungs, several factors affect the rate of gas diffusion across alveolar (the alveoli are tiny air sacs in the lungs) and tissue membranes. One of the factors is the difference in partial pressures of the gases across the membranes. The gases will diffuse from an area of high partial pressure to an area of low partial pressure, Look at the illustration below and note which blood vessels carry blood away from the lungs (arterial blood) and which blood vessels carry blood to the lungs (venous blood). The numbers on the diagram are the partial pressures of O₂ and CO₂ (mm Hg) in each of the areas shown. Pulmonary artery Right atrium Venous Blood Poog 46 mm Hg Vena Po: 40 mm Hg cavae Right ventricle oxygen-poor blood oxygen-rich blood Pulmonary Circuit Tissues Pco₂= 60 mm Hg Po: 30 mm Hg Name Lung Poog = 0.3 mm Hg 159 mm Hg Pog -Pulmonary vein 12-8 Left atrium Arterial blood 40 mm Hg Poz 100 mm Hg Aorta Po Left ventricle Use the data in the diagram above to explain why oxygen diffuses from the alveoli into the blood and from the blood into the tissues of the body. Use the data in the diagram above to explain why carbon dioxide diffuses from the tissues into the blood and from the blood into the alveoli and then finally out into the atmosphere. 14. In certain lung ailments such as emphysema, there is a decrease in the ability of oxygen to diffuse into the blood. How would the partial pressure of oxygen in the blood change? Why does a person with emphysema sometimes use a portable oxygen tank? Work Check 2. A 25.0 g sample of nitrogen gas, N₂, has a volume of 50.0 L and a pressure of 635 mm Hg. What is the temperature of the gas? Work Check 3. How many molecules of gas will fill a 30.0 gallon trash bag at 25 °C and 760. mm Hg? (1 L = 1.06 qts; 1 gal = 4 qts) 4. Aerosol containers can be dangerous if they are heated because they can explode. Suppose a container of hair spray with a pressure of 4.0 atm at a room temperature of 25 °C is thrown into a fire. a) If the temperature of the gas inside the aerosol can reaches 402 °C, what will the pressure be? b) The aerosol container may explode if the pressure inside exceeds 8.0 atm. Would you expect it to explode? 12-5 Wok Check Name 5. A volume of 500. mL of air on a cold winter day at 5 °C is inhaled into the lungs. The body temperature is 37 °C. What will be the new volume of the air in the lungs? Assume the pressure remains constant at 760 mm Hg. 6. The volume of air in a person's lungs is 615 mL at a pressure of 760. mm Hg. Inhalation occurs as the pressure in the lungs drops to 752 mm Hg. To what volume did the lungs expand? 7. A sample of gas containing 1.50 moles of neon has a volume of 8.50 L. What will be the volume of the sample of gas if a leak allows one-half of the neon atoms to escape? 8. A sample containing 4.80 g of oxygen gas, O₂, has a volume of 15.0 L. Oxygen gas is released until the volume is 10.0 L. How many moles of oxygen are removed? 12-6 C Name 9. What volume will 3.5g of CO occupy at 17 °C and 1875 torr? 10. A typical air sample in the lungs contains oxygen at 100 mm Hg, nitrogen at 573 mm Hg, carbon dioxide at 40 mm Hg, and water vapor at 47 mm Hg. Why are these pressures called partial pressures? 11. An anesthetic consists of a mixture of cyclopropane gas, C₂H₁, and oxygen gas, O₂. If the mixture has a total pressure of 825 torr, and the partial pressure of the cyclopropane gas is 73 torr, what is the partial pressure of the oxygen in the anesthetic? 12. If 74.5 % of exhaled air is nitrogen, what is the partial pressure of nitrogen in exhaled air. Assume that the total pressure of exhaled air is 760. mm Hg. 12-7 Work Check 13. During gas exchange in the lungs, several factors affect the rate of gas diffusion across alveolar (the alveoli are tiny air sacs in the lungs) and tissue membranes. One of the factors is the difference in partial pressures of the gases across the membranes. The gases will diffuse from an area of high partial pressure to an area of low partial pressure, Look at the illustration below and note which blood vessels carry blood away from the lungs (arterial blood) and which blood vessels carry blood to the lungs (venous blood). The numbers on the diagram are the partial pressures of O₂ and CO₂ (mm Hg) in each of the areas shown. Pulmonary artery Right atrium Venous Blood Poog 46 mm Hg Vena Po: 40 mm Hg cavae Right ventricle oxygen-poor blood oxygen-rich blood Pulmonary Circuit Tissues Pco₂= 60 mm Hg Po: 30 mm Hg Name Lung Poog = 0.3 mm Hg 159 mm Hg Pog -Pulmonary vein 12-8 Left atrium Arterial blood 40 mm Hg Poz 100 mm Hg Aorta Po Left ventricle Use the data in the diagram above to explain why oxygen diffuses from the alveoli into the blood and from the blood into the tissues of the body. Use the data in the diagram above to explain why carbon dioxide diffuses from the tissues into the blood and from the blood into the alveoli and then finally out into the atmosphere. 14. In certain lung ailments such as emphysema, there is a decrease in the ability of oxygen to diffuse into the blood. How would the partial pressure of oxygen in the blood change? Why does a person with emphysema sometimes use a portable oxygen tank? Work Check
Expert Answer:
Answer rating: 100% (QA)
2 The temperature of a gas can be calculated using the Ideal Gas Law PV nRT where P is pressure V is volume n is the number of moles R is the ideal gas constant and T is the temperature in kelvin Firs... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How simple or easy would you expect it to be to select managers for leadership and development at this company?
-
On a cold winter day you buy roasted chestnuts from a street vendor. Into the pocket of your down parka you put the change he gives youcoins constituting 9.00 g of copper at -12.0C. Your pocket...
-
What is the temperature inside an ideal refrigerator freezer that operates with a COP = 7.0 in a 22C room?
-
A firm has total debt of $6,000,000 and stockholder's equity is $4,000,000. The firm wants to calculate equity-to- total asset ratio in order to make decision about further raise of capital. What is...
-
Sylvia has received a $500,000 inheritance from her favorite, recently deceased aunt in Hawaii. Sylvia is planning to purchase a condo in Hawaii in the same area where her aunt lived all her life and...
-
Explain the concept of hop count in RIP. Can you explain why no hop is counted between N1 and R1 in Figure 20.15? Figure 20.15 Hop counts in RIP NI N2 N3 N4 Source Destination RI R2 R3 1 hop (N4) 2...
-
Timothy Muse opened a law office on October 2, 2010. During the first month of operations, the business completed the following transactions: Requirements 1. Open the following T-accounts: Cash,...
-
Filer manufacturing has 8.3 million shares of common stock outstanding. The current share price is $53 and the book value per share is $4. Filer manufacturing also has two bond issues outstanding....
-
9 . A cylindrical rod of diameter 5 cm and length 3 0 cm is exposed to a radiant heat flux of 2 0 0 W / m 2 . The rod is made of a material with a thermal conductivity of 5 0 W / mK and an emissivity...
-
Use the superposition principle to find io and vo in the circuit Fig. 4.79? Figure 4.79 For Prob. 4.11. 10 10 20 6 A 40 4 +30 V 10
-
The Kitchener Rangers are a junior ice hockey team that is a part of the Ontario Hockey League. They have called the Dom Cardillo arena at The Aud (located at 400 East Ave, Kitchener, ON N2H 1Z6)...
-
In this question you will consider the performance of a modified jet-propulsion cycle, for a turbojet engine with a normal-shock inlet, such as equipped on the Commonwealth Aircraft Corporation F86...
-
During the current month, Carla Vista Corporation completed Job 310 and Job 312. Job 310 cost $67,200 and Job 312 cost $48,000. Job 312 was sold on account for $86,400. Journalize the entries for the...
-
McWherter Instruments sold $600 million of 6% bonds, dated January 1, on January 1, 2024. The bonds mature on December 31, 2043 (20 years). For bonds of similar risk and maturity, the market yield...
-
The spot Dollar-GBP sterling exchange rate is $1.80 per pound. The U.S. and U.K. 1-year simple interest rates are 4% and 2% respectively. The futures price for delivery in one year is $1.86 per...
-
Briefly describe the forensic psychology career field Explain what drew you to this field (forensic psychology) Examine the impact of past, current, and developing technology in your field (forensic...
-
Harry wants to buy a new electric saw. In the shop there is a model with a recommended retail price (RRP) of $79.75 (including GST) but the salesman advises that all saws are on special and a 10%...
-
Write an essay describing the differing approaches of nursing leaders and managers to issues in practice. To complete this assignment, do the following: 1. Select an issue from the following list:...
-
A 71.2-g sample of oxalic acid, H2C2O4, was dissolved in 1.00 L of solution. How would you prepare 1.00 L of 0.150 M H2C2O4 from this solution?
-
Two elements in Period 3 are adjacent to one another in the periodic table. The ground-state atom of one element has only s electrons in its valence shell; the other one has at least one p electron...
-
Use Lewis symbols to represent the transfer of electrons between the following atoms to form ions with noble-gas configurations: a. Ca and Br b. K and I
-
The W10 \(\times 15\) cantilever beam is made of A-36 steel and is subjected to the loading shown. Determine the slope and displacement at its end \(B\). A 3 kip/ft 6 ft. -6 ft B
-
The two bars are pin connected at \(D\). Determine the slope at \(A\) and the displacement at \(D\). \(E I\) is constant. B 212 L2
-
Determine the slope at \(B\) and displacement at \(C . E I\) is constant. W W C02 312

Study smarter with the SolutionInn App


