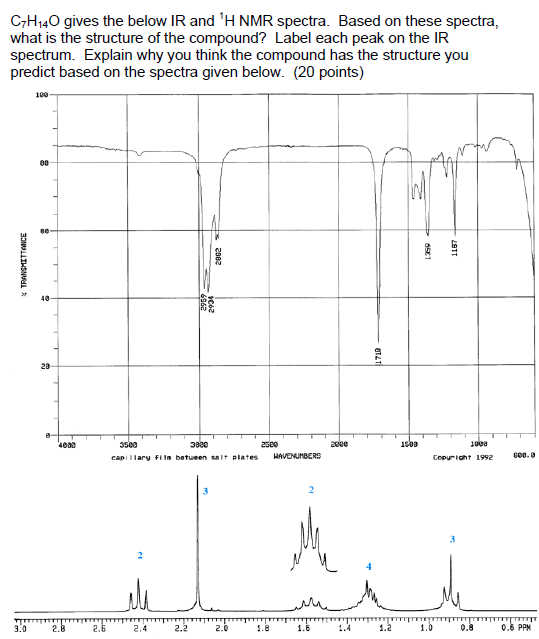
C7H40 gives the below IR and H NMR spectra. Based on these spectra, what is the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
C7H₁40 gives the below IR and ¹H NMR spectra. Based on these spectra, what is the structure of the compound? Label each peak on the IR spectrum. Explain why you think the compound has the structure you predict based on the spectra given below. (20 points) * TRANSMITTANCE TTTT 3.0 100- 80 28 4082 2.8 2.6 2.4 2934 3500 3800 capillary film between salt plates 2.2 2862 2.0 2500 HAVENUMBERS 2 2000 ST&T 1.2 르 1500 1359 1.0 1167 1000 Copyright 1992 0.8 600.0 0.6 PPM C7H₁40 gives the below IR and ¹H NMR spectra. Based on these spectra, what is the structure of the compound? Label each peak on the IR spectrum. Explain why you think the compound has the structure you predict based on the spectra given below. (20 points) * TRANSMITTANCE TTTT 3.0 100- 80 28 4082 2.8 2.6 2.4 2934 3500 3800 capillary film between salt plates 2.2 2862 2.0 2500 HAVENUMBERS 2 2000 ST&T 1.2 르 1500 1359 1.0 1167 1000 Copyright 1992 0.8 600.0 0.6 PPM
Expert Answer:
Answer rating: 100% (QA)
The structure of the compound based on the IR and H NMR spectra is propylbenzene The IR spectrum has ... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Explain in three paragraphs why you think mobile commerce has developed more rapidly in Asia than in US. Please include References
-
Explain why you think that task control or results control would be more appropriate in the setting that you have chosen. Do not use an example from the text.
-
Explain why you think goodwill is referred to as an unidentiable intangible asset. What types of items would be considered identiable intangible assets ? Is there any difference in the annual...
-
Modify Lookup to make a program LookupAndPut that allows put operations to be specified on standard input. Use the convention that a plus sign indicates that the next two strings typed are the...
-
Explain why the concept of an organization as an iceberg is important.
-
In Insight 5.2, we discussed how, because of the force of gravity from the Moon, the Earth moves in an orbit around a point that lies between it and the Moon. (a) Find the radius of the Earths orbit....
-
The November cash records of Stenback, Inc., follow: Stenbacks Cash account shows the balance of $6,172 on November 30. On November 30, Stenback received the following bank statement: Additional data...
-
The Blazon Manufacturing Companys costing system has two direct- cost categories: direct materials and direct manufacturing labor. Manufacturing overhead (both variable and fixed) is allocated to...
-
Provide an example of a system of your choice: 1 Discuss design criteria of the system chosen. (8) 2 Discuss the steps involved when initiating formal design changes of the chosen system. (10) 3 How...
-
The production manager at a factory manufacturing four types of light fittings (A, B, C and D) on an automated machine is fixing the schedule for the next week on this machine. HEIJUNKA SCHEDULING OF...
-
The speed u of a car is governed by the two equations (after rescaling) du f-v dt df 2f+2 tanhu. dt where f is the traction force produced by the engine and u is the throttle position. (a) Obtain a...
-
Milea Incorporated experienced the following events in Year 1, Its first year of operations: 1. Received $12,000 cash from the issue of common stock. 2. Performed services on account for $45,000. 3....
-
Cost of Goods Manufactured and Sold LeMans Company produces specialty papers at its Fox Run plant. At the beginning of June, the following information was supplied by its accountant: Direct materials...
-
An aqueous solution of NaOH contains 20% NaOH by mass. It is desired to produce a 12% NaOH solution by diluting the concentrated solution stream with a stream of pure water. Find the amount of pure...
-
You are making equal annual payments on a 5-year, 9% loan of $80,000. How much total interest will be paid over the entire 5-year life of the loan? Please answer using a financial calculator!
-
A cart with a mass of 4 kg is placed on an incline at an angle of 4 degrees. Assuming the cart is stationary on this incline, what static coefficient of friction is required if the static force is 11...
-
A company sells two models of a productbasic and premium. The basic model has a contribution margin per unit of $25 and unit sales of 750. The premium model has a contribution margin per unit of $40...
-
A business had revenues of $280,000 and operating expenses of $315,000. Did the business (a) Incur a net loss (b) Realize net income?
-
What products would you expect from the following reactions? (a) (b) (c) (d) (e) OMe OMe
-
1-Bromobicyclo [2.2.1] heptane does not undergo elimination (below) when heated with a base. Explain this failure to react. (Construction of molecular models may help.) Br
-
If we examine Table 21.1, we find that the methylphenols (cresols) are less acidic than phenol itself. For example, This behavior is characteristic of phenols bearing electron-releasing groups....
-
During its first year of operations, Environmental Products Inc. had sales of \(\$ 875,000\), all on account. Industry experience suggests that Environmental Products's bad debt expense will be \(\$...
-
Use the solution to exercise S4-8 to answer these questions about Environmental Products Inc. for 2020. 1. Start with Accounts Receivable's beginning balance \((\$ 80,000)\) and then post to the...
-
If Maritimes Ltd. fails to make an adjusting entry for the accrued interest, which of the following will happen a. Net income will be understated, and liabilities will be overstated. b. Net income...

Study smarter with the SolutionInn App


