Question: Calculate direct Labour QUESTION 4 (11.5 MARKS) Jade Sdn. Bhd. (JSB) producing and selling garden decoration in the Lembah Kelang area. the following are information
Calculate direct Labour
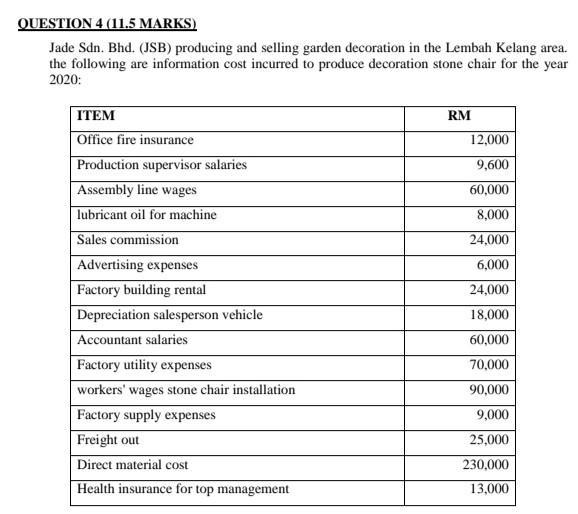
QUESTION 4 (11.5 MARKS) Jade Sdn. Bhd. (JSB) producing and selling garden decoration in the Lembah Kelang area. the following are information cost incurred to produce decoration stone chair for the year 2020: ITEM Office fire insurance Production supervisor salaries Assembly line wages lubricant oil for machine Sales commission Advertising expenses Factory building rental Depreciation salesperson vehicle Accountant salaries Factory utility expenses workers' wages stone chair installation Factory supply expenses Freight out Direct material cost Health insurance for top management RM 12,000 9,600 60,000 8,000 24,000 6,000 24,000 18,000 60,000 70,000 90,000 9,000 25,000 230,000 13,000
Step by Step Solution
3.53 Rating (160 Votes )
There are 3 Steps involved in it
Direct labour cost is the cost incurred for the wages of the workers who are dire... View full answer

Get step-by-step solutions from verified subject matter experts


