Calculate E for the following reactions at 298 K. Ag(NH3)+ Ag + 2NH3 Given: Ag(CN)2+eAg...
Fantastic news! We've Found the answer you've been seeking!
Question:

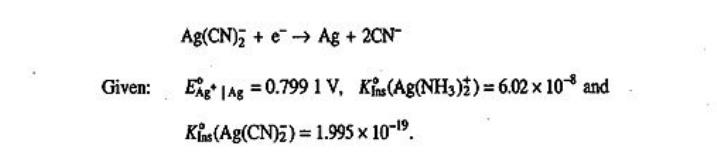
Transcribed Image Text:
Calculate E for the following reactions at 298 K. Ag(NH3)+ →→ Ag + 2NH3 Given: Ag(CN)2+eAg + 2CN Eg | Ag = 0.799 1 V, Kins(Ag(CN)₂) = 1.995 × 10-1⁹. Kins (Ag(NH3)2) = 6.02 x 10 and Calculate E for the following reactions at 298 K. Ag(NH3)+ →→ Ag + 2NH3 Given: Ag(CN)2+eAg + 2CN Eg | Ag = 0.799 1 V, Kins(Ag(CN)₂) = 1.995 × 10-1⁹. Kins (Ag(NH3)2) = 6.02 x 10 and
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate DG for the following reactions at 25C: (a) N2(g) + O2(g) 2NO(g) (b) H2O(l) H2O(g) (c) 2C2H2(g) + 5O2(g) 4CO2(g) + 2H2O(l)
-
Calculate DG for the following reactions at 25C: (a) 2Mg(s) + O2(g) 2MgO(s) (b) 2SO2(g) + O2(g) 2SO3(g) (c) 2C2H6(g) + 7O2(g) 4CO2(g) + 6H2O(l) See Appendix 3 for thermodynamic data.
-
Calculate DG and Kc for the following reactions at 25C: (a) (b) (c) (d) 2 + Br2() 21 (aq)2Br (aq) I2(s) 02(8) 4H (a) 4Fe (aq) 2H20(1) + 4Fe (aq) 2.A l (s) + 312(s) 2A13+(aq) + 6(aq)
-
Calculate the following limits at infinity. If necessary, use -inf and inf to denote -co and co. Use DNE if a limit does not exist. 1. lim e = 818 2. lim e = 8178 3. lim In(x) 24x In(x) =
-
Talk to someone who regularly interviews candidates for entry-level jobs. Possible questions to ask include the following: How long have you been interviewing for your organization? Does everyone on...
-
In 2005, General Motors announced a 20 percent reduction in its staffing levels and the closure of many assembly plants. Those laid off as a result would likely be classified as a. Seasonally...
-
Two \(0.500-\mathrm{kg}\) carts are \(100 \mathrm{~mm}\) apart on a low-friction track. You push one of the carts with a constant force of \(2.00 \mathrm{~N}\) directed so that the cart you push...
-
A lighting company seeks to study the percentage of defective glass shells being manufactured. Theoretically, the percentage of defectives is dependent on temperature, humidity, and the level of...
-
Advise the company on strategies that in your opinion should be used by the company (Do you think that they are promoting their products correctly? Is there anything else that they could do- suggest...
-
In alphabetical order below are balance sheet items for Mendoza Company at December 31, 2017. Kathy Mendoza is the owner of Mendoza Company. Prepare a balance sheet, following the format of...
-
Employee Payroll Using the table below, create the paycheques 705-709 for the pay period ending January 14, 2021. Use the following information. Perform a back up, then use the History and select...
-
If vector vec (v)=vec (i)-2vec (j) and vector vec (w)=3vec (i)-3vec (j), find the magnitude of vector (2vec (v)-3vec (w)). Leave your answer in simplest radical form.
-
Consider the following data: Scenario Probability Return K Return K WI W2 0.1 0.4 40% 10% 10% 5% 0.5 -10% -2% Wa Let V(t)=xSi(t) + x2S2(t) (a) Assuming the initial weighting is 50% invested in S,...
-
Carolyn Bennett would like you to analyze the potential financial impact of these forecasting models. Based on the data of the actual demand at Coors Field, and the estimates of each model, we can...
-
Two types of football helmets, padded and suspension were compared to determine which had the lowest rate of impact damage. Forty helmets of each type were randomly selected and subjected to an...
-
The equilibrium price for parking in the downtown area of a large city is about $35 a day, this is represented by Pe in the graph above. The city council is looking at ways to resolve parking...
-
You are the CFO of a retail company, VL Inc. You just closed Fiscal Year 2020 and completed the balance sheets, income statements, and statement of cashflows. Company CEO, Lee, is asking for a detail...
-
A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method to estimate the mole-fraction compositions of the distillate and bottoms. Assume initial overhead and...
-
In the flushing and cleaning of columns used in liquid chromatography, a series of solvents is used. Hexane (C6H14), chloroform (CHCl3), methanol (CH3OH), and water are passed through the column in...
-
The Ostwald process for the commercial production of nitric acid from ammonia and oxygen involves the following steps: 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g) 2NO(g) + O2(g) 2NO2(g) 3NO2(g) + H2O(l) ...
-
The high melting points of ionic solids indicate that a lot of energy must be supplied to separate the ions from one another. How is it possible that the ions can separate from one another when...
-
Frieda Wannamaker is a taxable investor who is currently in the 28% income-tax bracket. She is considering purchasing a tax-exempt bond with a yield of 3.75%. The taxable equivalent yield on this...
-
Technical analysis looks at the demand and supply for securities based on trading volumes and price studies. Charting is a common method used to identify and project price trends in a security. A...
-
A rationale for the use of the price-to-sales (P/S) approach is: a. Sales are more volatile than earnings. b. P/S ratios assess cost structures accurately. c. Revenues are less subject to accounting...

Study smarter with the SolutionInn App



