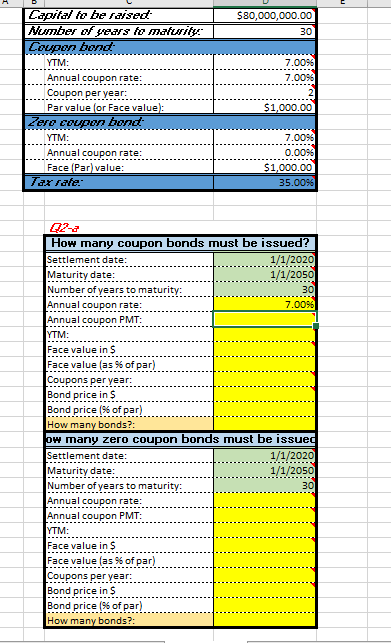
calculate hoW many Coupon bond must be Issued? calculate hoW many Zero Coupon bond must be Issued?
Fantastic news! We've Found the answer you've been seeking!
Question:
calculate hoW many Coupon bond must be Issued?
calculate hoW many Zero Coupon bond must be Issued?

Related Book For 

Foundations of Financial Management
ISBN: 978-1259024979
10th Canadian edition
Authors: Stanley Block, Geoffrey Hirt, Bartley Danielsen, Doug Short, Michael Perretta
Posted Date:




