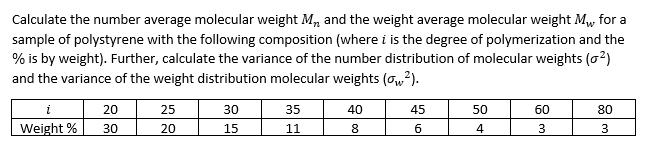
Calculate the number average molecular weight M, and the weight average molecular weight M, for a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculate the number average molecular weight M, and the weight average molecular weight M₁, for a sample of polystyrene with the following composition (where i is the degree of polymerization and the % is by weight). Further, calculate the variance of the number distribution of molecular weights (0²) and the variance of the weight distribution molecular weights (ow²). i 20 25 30 35 40 45 50 60 80 Weight % 30 20 15 11 8 6 4 3 3 Calculate the number average molecular weight M, and the weight average molecular weight M₁, for a sample of polystyrene with the following composition (where i is the degree of polymerization and the % is by weight). Further, calculate the variance of the number distribution of molecular weights (0²) and the variance of the weight distribution molecular weights (ow²). i 20 25 30 35 40 45 50 60 80 Weight % 30 20 15 11 8 6 4 3 3
Expert Answer:
Answer rating: 100% (QA)
Degree of Polymerization DP molecular weight mer weig... View the full answer

Related Book For 

Statistics for the Life Sciences
ISBN: 978-0321989581
5th edition
Authors: Myra Samuels, Jeffrey Witmer, Andrew Schaffner
Posted Date:
Students also viewed these chemistry questions
-
The following histogram shows the distribution for a sample of data:
-
Calculate the time required for a sample of radioactive tritium to lose of its activity. (Tritium has a half-life of 12.3 years.)
-
For a sample of 36 houses, what would you expect the distribution of the sale prices to be? A real-estate agent has been assigned 10 houses at random to sell this month. She wants to know whether the...
-
The Chief Financial Officer at Ford Motor Company is said to usea hybrid-costing system. Define the hybrid-costing system. Explainthe advantages to this company to use this system. I want a 10 page 2...
-
Explain which approach, market concentration or market spreading, should be the primary strategy used in the following situations:
-
Provide an example for each component of the hyperpersonal perspective and determine whether or not the approach merits modification.
-
In the region just downstream of a sluice gate, the water may develop a reverse flow region as is indicated in Fig. P4.59 and Video V10.9. The velocity profile is assumed to consist of two uniform...
-
Marthas Wonderful Cookie Company makes a special super chocolate-chip peanut butter cookie. The company would like the cookies to average approximately eight chocolate chips apiece. Too few or too...
-
Could you please give me answers that are supported by references? There are some acts that are considered to be criminal in nature based on the likelihood of creating a disturbance to the peace or...
-
Shawn and Amy were college sweethearts and had been married for 20 wonderful years. They lived in Denver, Colorado. Shawn was one of three partners with the OMG! Engineering firm. Unfortunately,...
-
The following information is for Cullumber Ltd in July Cash balance per bank July 31, $10,770 Cash balance per books, July 31, S8,965 Bank service charge. $50 Deposits in transit. $1.937 5. 1. 2....
-
Who is a great example of a Transactional Leadership and Laissez-Faire Leadership in the 20 century?
-
How might the consequences of the situation have been different if acted as a Creator?
-
About costs incurred after the acquisition, what does increased service potential mean?
-
Define the significant differences and similarities between empowerment and laissez-faire leadership.?
-
If you were the leader of your organization, how would you change the way your organization communicates in order to assure that all employees contribute to the organization's mission, vision, and...
-
A high-tech machine that produces watch bands costs $849,000. This cost could be depreciated at 30% per year (CCA Class 10). The machine would be worth $175,000 in five years. There are no capital...
-
According to a New York Times columnist, The estate tax affects a surprisingly small number of people. In 2003, . . . just 1.25 percent of all deaths resulted in taxable estates, with most of them...
-
Data from a large study indicate that the serum concentration of lactate dehydrogenase (LD) is higher in men than in women. (The data are summarized in Example 7.6.1.) Suppose Dr. Sanchez proposes to...
-
Consider the data from Exercise 11.S.5. For these data, SS(ozone) = 0.696, SS(sulfur) = 0.492, SS(interaction) = 0.166, and SS(within) = 0.275. (a) Prepare an interaction graph (like Figure 11.7.3)....
-
Proceed as in Exercise 11.2.1 for the following data: (a) Compute SS(between) and SS(within). (b) Compute SS(total), and verify the relationship between SS(between), SS(within), and SS(total). (c)...
-
Suppose a bond is taxable for both federal and state purposes. Let Rb = the BTROR on the bond, tfed = the federal tax rate, and tst = the state tax rate. Determine the ATROR (i.e., after federal and...
-
Under what conditions are the Exempt Model and Pension Model equivalent? Under what conditions would one model perform better than the other? How does the \($6,500\) limitation on deductible and Roth...
-
Consider three taxpayers who are in the following tax brackets: Alice 25% Brad 35% Camille 40%

Study smarter with the SolutionInn App


