Calculate the change in the standard entropy of the system, ASO, for the synthesis of ammonia...
Fantastic news! We've Found the answer you've been seeking!
Question:
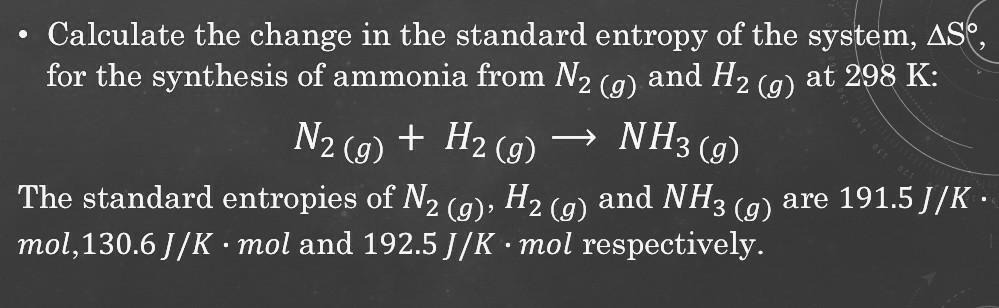
Transcribed Image Text:
Calculate the change in the standard entropy of the system, ASO, for the synthesis of ammonia from N₂ (g) and H₂ NH3 (9) (g) at 298 K: N2 (g) + H₂(g) The standard entropies of N₂ (g), H₂ (g) and NH3 (g) are 191.5 J/K · mol,130.6J/K · mol and 192.5 J/K mol respectively. ● Calculate the change in the standard entropy of the system, ASO, for the synthesis of ammonia from N₂ (g) and H₂ NH3 (9) (g) at 298 K: N2 (g) + H₂(g) The standard entropies of N₂ (g), H₂ (g) and NH3 (g) are 191.5 J/K · mol,130.6J/K · mol and 192.5 J/K mol respectively. ●
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the change in the entropies of the system and the surroundings, and the total change in entropy, when the volume of a sample of argon gas of mass 21 gat 298 K and 1.50 bar increases from...
-
Calculate the change in entropy of 250 g of water heated slowly from 20.0C to 80.0C. (Suggestion: Note that dQ = mc dT)
-
Calculate the change in entropy when 50 k] of energy is transferred reversibly and isothermally as heat to a large block of copper at (a) OC, (b) 70e
-
Rio Tinto is a listed company in the mining and metals production industry. Some information drawn from its 2020 annual report is shown below. Safety and health performance 2020 2019 2018 Fatalities...
-
Compare and contrast the relative advantages and disadvantages of sequential, block, group, alphabetic, and mnemonic codes.
-
Everyday experience indicates that it is much easier to compress gases than liquids. This property is measured by the isothermal compressibility, =1/V V/p| T , the fractional change in a substances...
-
Refer to the bond situation of Hewitt Corporation in Exercise 15-27. Hewitt issued the bonds at the advertised price. The company uses the straight-line amortization method and reports financial...
-
1. Describe the mechanisms that WorldComs management used to transfer profit from other time periods to inflate the current period. 2. Why did Arthur Andersen go along with each of these mechanisms?...
-
When car has a lifetime distribution exp(1/4), the owner wants to change the car either it is broken or the car is 8 years old. If it is replaced at 8 years (not broken), the owner pays 20, 000...
-
Jackson Manufacturing holds a 150-day note of $10,000 that has an interest rate of 7% annually. After 90 days, the note is sold to a bank at a discount rate of 6% annually. Find the proceeds on the...
-
Pots purchased 70% of Spatula's common shares for $210,000 on June 1, Year 4. Pots and Spatula Inc. had the following balance sheets on June 1, Year 4 prior to the purchase: Pots Spatula Fair Value...
-
A circuit with a total charge of 81 C is shown below. Calculate the energy if C = 3.0 F. Express your answer with the appropriate units. U = HA Value ? Units C 8V
-
A Break-Even Analysis is best used as a preliminary planning tool. We can use break-even analysis to examine theopportunity cost of investing in the equipment as a capital expense, as opposed to...
-
5. Write a program to print multiplication tables of numbers. Each number whose table is to be printed should be a thread instance of different thread class. Thus, you will create two classes...
-
Describe in steps how to determine the strengths and weaknesses of a research study? Also provide reference or links.
-
If offshore assembly provisions were extended to include more goods, what would this do to the actual level of protection provided by a country's nominal tariff schedule? Explain. If the extension of...
-
lease provide the detailed answer for assignment. TIA. The diagram is important, kindly help to draw and provide. During process discovery, you interview the process owner, who provides high-level...
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
Show that the volume of a triclinic unit cell of sides a, b, and c and angles a, B3, and y is V= abc (1- cos2a- cos2B cos2y+ 2 cos a cos B cos y)1/2 Use this expression to derive expressions for...
-
Find an expression for the fugacity coefficient of a gas that obeys the equation of state pVm = RT(1 +B/Vm + C/V-1). Use the resulting expression to estimate the fugacity of argon at 1.00 am3 and 100...
-
Estimate the coefficients a and b in the Dieterici equation of state from the critical constants of xenon. Determine pressure exerted by 1.0 mol Xe when it is confined to 1.0 dm3 at 25C.
-
Brown & Brown, CPAs, was engaged by the board of directors of Cook Indus tries, Inc. to audit Cooks calendar year 19X5 financial statements. The following report was drafted by an audit assistant at...
-
Your client, S Company is a major manufacturer of widgets. Your audit report for the previous year was unqualified. During the current year, several class- action suits were filed against the company...
-
Q Company is a gas distribution company that is regulated by the state public service commission. The commission must approve all rate changes made by Q Company, but Q Company is allowed to collect...

Study smarter with the SolutionInn App



