13 The graph shows the variation in concentrations of the three gases present in a mixture...
Fantastic news! We've Found the answer you've been seeking!
Question:
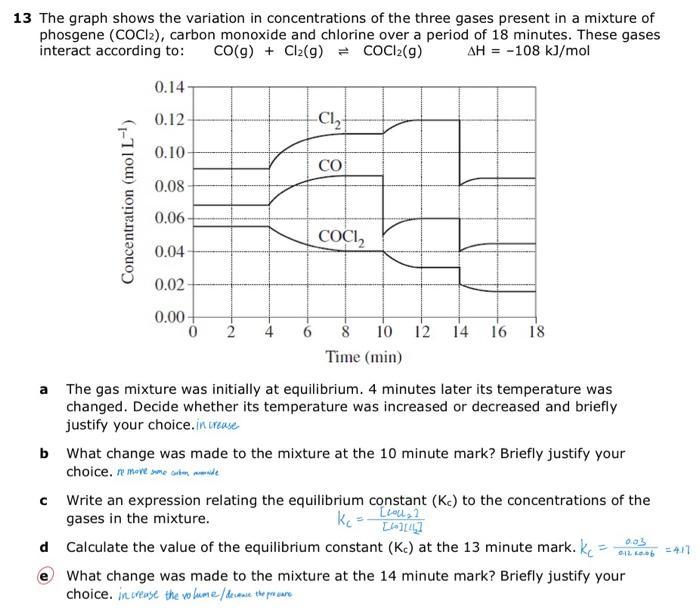
Transcribed Image Text:
13 The graph shows the variation in concentrations of the three gases present in a mixture of phosgene (COCI2), carbon monoxide and chlorine over a period of 18 minutes. These gases interact according to: CO(g) + Cl₂(g) = COCl2(g) AH = -108 kJ/mol Concentration (mol L-¹) 0.14- 0.12- 0.10 0.08- 0.06- 0.04- 0.02- 0.00+ 0 2 -C₁₂ CO COCI₂2 10 12 14 16 18 Time (min) a The gas mixture was initially at equilibrium. 4 minutes later its temperature was changed. Decide whether its temperature was increased or decreased and briefly justify your choice.increase b What change was made to the mixture at the 10 minute mark? Briefly justify your choice. remove me tende с Write an expression relating the equilibrium constant (Kc) to the concentrations of the gases in the mixture. kc [lol₂] [4][44] d Calculate the value of the equilibrium constant (Kc) at the 13 minute mark. k 0.03 012 60.06 What change was made to the mixture at the 14 minute mark? Briefly justify your choice. increase the volume / decause the procure = 4.17 13 The graph shows the variation in concentrations of the three gases present in a mixture of phosgene (COCI2), carbon monoxide and chlorine over a period of 18 minutes. These gases interact according to: CO(g) + Cl₂(g) = COCl2(g) AH = -108 kJ/mol Concentration (mol L-¹) 0.14- 0.12- 0.10 0.08- 0.06- 0.04- 0.02- 0.00+ 0 2 -C₁₂ CO COCI₂2 10 12 14 16 18 Time (min) a The gas mixture was initially at equilibrium. 4 minutes later its temperature was changed. Decide whether its temperature was increased or decreased and briefly justify your choice.increase b What change was made to the mixture at the 10 minute mark? Briefly justify your choice. remove me tende с Write an expression relating the equilibrium constant (Kc) to the concentrations of the gases in the mixture. kc [lol₂] [4][44] d Calculate the value of the equilibrium constant (Kc) at the 13 minute mark. k 0.03 012 60.06 What change was made to the mixture at the 14 minute mark? Briefly justify your choice. increase the volume / decause the procure = 4.17
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
When a firm needs to raise money via a bond issue, one of the quickest ways is through a______. This activity involves the purpose of a large block of securities by a large institutional investor...
-
Financial markets allow savers and borrowers to meet differing consumption preferences if Select one a. a limited number of financial intermediaries exist b. riskier borrowers are able to access...
-
The information in the data table above represents two different trials for an experiment to study the rate of the reaction between NO (g) and H 2(g) , as represented by the balanced equation above...
-
The following auditors report was drafted by a staff accountant of Turner & Turner, CPAs, at the completion of the audit of the financial statements of Lyon Computers, Inc. (a non-public company) for...
-
Mendenhall and Sincich (1993) present data that can be used to investigate allegations of gender discrimination in the hiring practices of a particular firm. These data are as follows: In this table,...
-
Valuation fundamentals Nelson wants to increase the production capacity of his cattle food plant in Pretoria, South Africa. He is trying to evaluate the economics of purchasing a pelleting machine...
-
Consider the following frequency distribution. Construct a cumulative frequency distribution. Bin Frequency 10-19 10 20-29 14 30-39 40-49 17 7 50-59 2
-
On January 1, 2010, Terrel Company sold $100,000 of 10-year, 8% bonds at 93.5, an effective rate of 9%. Interest is to be paid on July 1 and December 31. Compute the amount of premium or discount...
-
Control systems exist in numerous varieties, both in type of applications and level of sophistications. Since most systems are dynamic in nature, to control or regulate against a set of desired...
-
Adriaan Taylor Corporation is a newly formed entity that engages in the purchase and resale of amphibious tour vehicles. Purchases for the first year of operation were as follows: Date Purchases...
-
An automobile company partnered with its competition to compile a large sample of email contacts from which they sent questionnaires to a random sample that includes car owners who aren't necessarily...
-
Who is more powerful, the governor or the lieutenant governor? Why do you think this?
-
Provide some examples about the Human Resources management problem of Disneyland in China.
-
You are the Director of Human Resources at a city. You are about to begin contract negotiations of a labor contract. What others from the management staff do you ask to attend the negotiation...
-
what arte examples of cultural and social sensitivities in a work environment ?
-
When conducting a job analysis, what are the five methods are most effective?
-
Crystal Water, a community committed to raise awareness towards water pollution in big cities, permission approved to install CCC an innovation water cleaning device. Administrative costs to obtain...
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
A certain reaction is spontaneous at 72C. If the enthalpy change for the reaction is 19 kJ/mol, what is the minimum value of S (in J/K ? mol) for the reaction?
-
A compound has the empirical formula SF4. At 20C, 0.100 g of the gaseous compound occupies a volume of 22.1 mL and exerts a pressure of 1.02 atm. What is the molecular formula of the gas?
-
Ethanol, C2H5OH, and dimethyl ether, CH3OCH3, are structural isomers. Compare their melting points, boiling points, and solubilities in water.
-
Describe the five financial statements and how they are prepared.
-
Presented below in alphabetical order are statement of financial position items for Feagler Gardening at December 31, 2025. Prepare a statement of financial position following the format of...
-
Presented below is selected information related to Tsui Repairs at December 31, 2025. Tsui reports financial information monthly (amounts in thousands). a. Determine the total assets of Tsui Repairs...

Study smarter with the SolutionInn App


