The aim of this experiment is to determine the partition coefficient of succinic acid in system...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
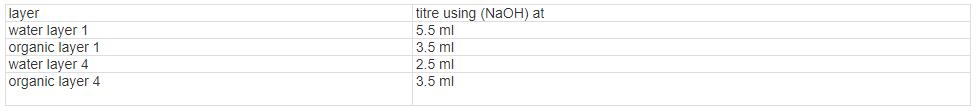
The aim of this experiment is to determine the partition coefficient of succinic acid in system of two immiscible liquids: 1-butanol - water. The measurement will be performed at 4 different concentrations of succinic acid and different volume ratios of partitioned mixture (1-butanol - water). CH₂-COOH CH₂-COOH Reaction for succinic acid and sodium hydroxide 1 16. MATERIALS 2024 = 80 - Succinic acid (aqueous solution c-20g/dm³) 0.05 M NaOH (500 mls) 1-butanol, Phenolphthalein PROCEDURE: Funnel No. 1 2 3- 2NaOH 3 4 1. Prepare mixtures of butanol, water and succinic acid in separation funnels as outlined in the table below. 3. 2. Gently shake the separating funnels with prepared solutions for 20 min. Allow mixtures to stand and separate into two clear layers: lower layer is water (Wat), and upper layer is butanol (Org). Pour carefully the water phase into a beaker using the funnel's valve. layers). Table: Composition of Mixtures. 1 4. For each mixture titrate 10 ml aliquots of the water phase against NaOH using phenolphthalein as indicator. Rem; All titrations must be repeated. 5. Repeat step 4 for the butanol phase (note; discard the interface between both. 4 (29)//250ml -50 m² go5x0-25=0.0125 mols. &=0.0125 x 40- (0.5 used 3g Succinic acid (ml) 40 20 30 20 CH2-COONa CH2-COONa water оля Wat I Water (ml) 30 . 20 + 2 H₂O 1-butanol (ml) 30 30 X 30 x 30 NaOH Titrat at 5.5ml 3.5 2.5 m 3.5 CALCULATIONS 1. Calculate the concentration of succinic acid in the water and organic phase, cWat, cOrg, respectively, for each composition of the partition mixture 2. Calculate the average partition coefficient k of succinic acid at each composition of thepartition mixture. K=[Succinic]wa/[ Succinic]org layer water layer 1 organic layer 1 water layer 4 organic layer 4 titre using (NaOH) at 5.5 ml 3.5 ml 2.5 ml 3.5 ml The aim of this experiment is to determine the partition coefficient of succinic acid in system of two immiscible liquids: 1-butanol - water. The measurement will be performed at 4 different concentrations of succinic acid and different volume ratios of partitioned mixture (1-butanol - water). CH₂-COOH CH₂-COOH Reaction for succinic acid and sodium hydroxide 1 16. MATERIALS 2024 = 80 - Succinic acid (aqueous solution c-20g/dm³) 0.05 M NaOH (500 mls) 1-butanol, Phenolphthalein PROCEDURE: Funnel No. 1 2 3- 2NaOH 3 4 1. Prepare mixtures of butanol, water and succinic acid in separation funnels as outlined in the table below. 3. 2. Gently shake the separating funnels with prepared solutions for 20 min. Allow mixtures to stand and separate into two clear layers: lower layer is water (Wat), and upper layer is butanol (Org). Pour carefully the water phase into a beaker using the funnel's valve. layers). Table: Composition of Mixtures. 1 4. For each mixture titrate 10 ml aliquots of the water phase against NaOH using phenolphthalein as indicator. Rem; All titrations must be repeated. 5. Repeat step 4 for the butanol phase (note; discard the interface between both. 4 (29)//250ml -50 m² go5x0-25=0.0125 mols. &=0.0125 x 40- (0.5 used 3g Succinic acid (ml) 40 20 30 20 CH2-COONa CH2-COONa water оля Wat I Water (ml) 30 . 20 + 2 H₂O 1-butanol (ml) 30 30 X 30 x 30 NaOH Titrat at 5.5ml 3.5 2.5 m 3.5 CALCULATIONS 1. Calculate the concentration of succinic acid in the water and organic phase, cWat, cOrg, respectively, for each composition of the partition mixture 2. Calculate the average partition coefficient k of succinic acid at each composition of thepartition mixture. K=[Succinic]wa/[ Succinic]org layer water layer 1 organic layer 1 water layer 4 organic layer 4 titre using (NaOH) at 5.5 ml 3.5 ml 2.5 ml 3.5 ml
Expert Answer:
Answer rating: 100% (QA)
Certainly Lets perform the calculations step by step and explain the process 1 Calculation of succin... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
Calculate the concentration of an aqueous solution of NaOH that has a pH of 11.50.
-
Calculate the concentration of an aqueous solution of Ca(OH)2 that has a pH of 10.05.
-
Calculate the concentration of the acid (or base) remaining in solution when 10.7 mL of 0.211 M HNO3 are added to 16.3 mL of 0.258 M NaOH.
-
Consider the market for milk. Draw a supply curve and a demand curve (is the demand curve elastic on inelastic). Label the equilibrium price and quantity. Suppose that the production of milk causes...
-
Rodriguez (2003) suggests when small businesses do actually launch new products, they can do so in half the time and effort a big firm takes. How?
-
A student on a distant planet performs a "loop-the-loop" experiment. She releases a frictionless 1.3 kg cart from a height of 4.50m. It is observed that the track exerts a downward, normal force of...
-
You borrow \($495\) from your brother-in-law to buy an iPad and agree to pay it back when you get your tax refund (6 months away). Because you are family, but not trusted, you are being charged...
-
Create a list of user requirements for the problem definition created in Problem 4.
-
Explain how gross domestic product is calculated using the income approach and the expenditure approach. Compare the classical economic theory that was used prior to the Great Depression to the...
-
Data table Buckland Associates Bank Reconciliation Cash February 28, 2025 Bank: Beg. Bal 4,995 650 Feb. 3 Balance, February 28, 2025 4.707 Feb 6 600 2200 Feb. 12 Add Deposit in transit 2.200 Feb. 15...
-
If Inverse Demand is given by the expression P = 100-2Q and Inverse Supply is given by the expression P = -20 + 3Q, what is the PED at the point where prices are 20 below the equilibrium?
-
The queue is implemented as a class containing an array of items, a data member indicating the index of the last item put on the queue (rear), a data member indicating the index of the location...
-
The stack is implemented as a class containing an array of items, a data member indicating the index of the last item put on the stack (top), and two Boolean data members, underFlow and overFlow. The...
-
The following output (from \(\mathrm{R}\) ) presents the results of a hypothesis test for a population mean \(\mu\). a. Is this a one-tailed or a two-tailed test? b. What is the null hypothesis? c....
-
What is the Big-O measure for initializing the free list in the arraybased linked implementation? For the GetNode and FreeNode functions?
-
Does a derived class have access to the private data members of the base class?
-
This report aims to address a real-life problem and propose a conceptual model to solve it. The report is divided into several sections, starting with a description of the problem, followed by the...
-
How do the principles of (a) Physical controls and (b) Documentation controls apply to cash disbursements?
-
From Equation 8.4 and the ionic radii given in Figure 7.7, calculate the potential energy of the following pairs of ions. Assume that the ions are separated by a distance equal to the sum of their...
-
What is the molecular formula of each of the following compounds? (a) Empirical formula CH2,molsr mass = 84g/mol (b) Empirical formula NH2Cl,molar mass = 51.5 g /mol
-
Calculate the molality of each of the following solutions: (a) 8.66 g benzene (C6H6) dissolved in 23.6 g carbon tetrachloride (CCl4), (b) 4.80 g NaCl dissolved in 0.350 L of water?
-
Consider a two-level system, with basis \(|1angle,|2angle\), and in this basis, a Hamiltonian with elements \(\left(\begin{array}{ll}1 & 1 \\ 1 & 1\end{array} ight)\). Use the first form of the...
-
Use the practical variational method for the same harmonic oscillator ground state energy, with trial wave function \(\psi_{a}(x)=e^{-a y^{2}}\).
-
Use the Ritz variational method for the harmonic oscillator, with trial wave functions \(\psi_{1}(x)=\) \(e^{-y^{2} / 2}, \psi_{2}(x)=e^{-y^{2}}, \psi_{3}(x)=e^{-2 y^{2}}\), where \(y=x \sqrt{m...

Study smarter with the SolutionInn App


