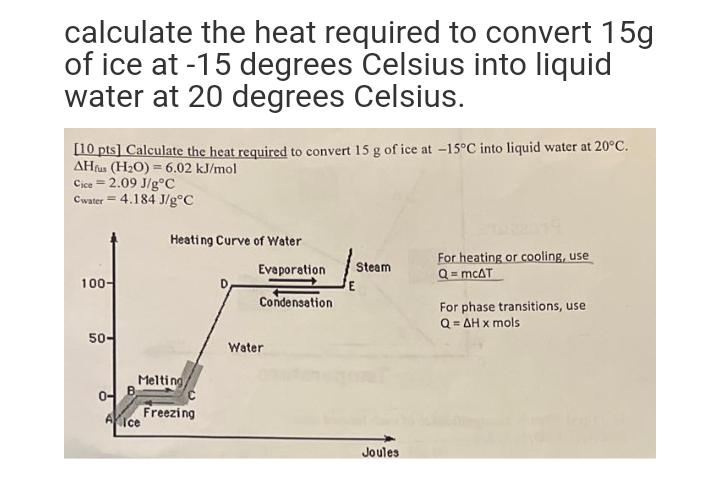
calculate the heat required to convert 15g of ice at -15 degrees Celsius into liquid water...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
calculate the heat required to convert 15g of ice at -15 degrees Celsius into liquid water at 20 degrees Celsius. [10 pts] Calculate the heat required to convert 15 g of ice at -15C into liquid water at 20C. AHfus (HO) = 6.02 kJ/mol Cice=2.09 J/gC Cwater = 4.184 J/gC 100- 50- 0- Heating Curve of Water Melting Aice Freezing Evaporation Condensation Water E Steam Joules For heating or cooling, use Q = mcAT For phase transitions, use Q=AH x mols calculate the heat required to convert 15g of ice at -15 degrees Celsius into liquid water at 20 degrees Celsius. [10 pts] Calculate the heat required to convert 15 g of ice at -15C into liquid water at 20C. AHfus (HO) = 6.02 kJ/mol Cice=2.09 J/gC Cwater = 4.184 J/gC 100- 50- 0- Heating Curve of Water Melting Aice Freezing Evaporation Condensation Water E Steam Joules For heating or cooling, use Q = mcAT For phase transitions, use Q=AH x mols
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The heat required to raise the temperature from 300.0 K to 400.0 K for 1 mole of a gas at constant volume is 2079 J. The internal energy required to heat the same gas at constant pressure from 550.0...
-
Calculate the heat required to raise 50 kg of solid sodium carbonate (Na2CO3) from 10C to 50C at 1atm using (a) The true heat capacity of Na2CO3, which is 1.14 kJ / (kg C). (b) A heat capacity...
-
Estimate the heat required to heat a 0.15-kg apple from 12 C to 36 C. (Assume the apple is mostly water.)
-
Provide an overview of the OS and the manufacturer What is the footprint for the selected OS? What security architecture was implemented? How many CVEs does this OS have?
-
What is product liability? Explain the charges that most often form the basis for product liability claims. What must a customer prove under these charges?
-
Barlow Company manufactures three products A , B , and C . The selling price, variable costs, and contribution margin for one unit of each product follow: Product A B C Selling price $ 1 8 0 $ 2 4 0...
-
What are the four concepts that are essential to the extended object-oriented approach to software development?
-
A firm reported comprehensive income of $376 million for 2009, consisting of $500 million in operating income (after tax) less $124 million of net financial expenses (after tax). It also reported the...
-
what is a strong-form/semi strong-form/not strong form marketing? whats their influences on purchasing stock?
-
A collar B, of mass m and negligible dimensions, is attached to the rim of a hoop of the same mass m and of radius r that rolls without sliding on a horizontal surface. Determine the angular velocity...
-
How do sociological theorists conceptualize the intersectionality of social structures and individual agency, and to what extent does this understanding inform strategies for societal transformation...
-
How are guard conditions shown on a behavioral state machine?
-
14.27 Let \(\zeta\) denote a generic measure of association. For \(K\) independent multinomial samples of sizes \(\left\{n_{k} ight\}\), suppose that \(\sqrt{n_{k}}\left(\hat{\zeta}_{k}-\zeta_{k}...
-
In Problem 126, if Juan decided to wait 10 years before investing for retirement, how much would he have to invest on his thirty-sixth birthday to have the same account balance on his sixtieth...
-
Show that the zeroth polyphase component of an \(L\) th band filter is constant in the frequency domain.
-
Show that, for a realization of a WSS process \(\{X\}\) applied as input process to a serial-to-parallel converter, the PSD matrix \(\Gamma_{X}(z)\) is pseudo-circulant.
-
Garrett Group is financed with 40% debt, 10% preferred stock, and 50% equity. It pays a 9% coupon on its bonds, which have a YTM of 6%. Its preferred stock pays an annual dividend of $4 and sells...
-
In order to get an idea on current buying trends, a real estate agent collects data on 10 recent house sales in the area. Specifically, she notes the number of bedrooms in each house as follows: a....
-
For orbital that are symmetric but not spherical, the contour representations (as in Figures 6.22 and 6.23) suggest where nodal planes exist (that is, where the electron density is zero). For...
-
(a) How is the concept of effective nuclear charge used to simplify the numerous electron-electron repulsions in a many electron atom? (b) Which experiences a greater effective nuclear charge in a Be...
-
The reaction 2 Mg(s) + O2(g) 2 MgO(s) is highly spontaneous and has a negative value for . The second law of thermodynamics states that in any spontaneous process there is always an increase in the...
-
The pendulum on a grandfather clock has a period of 2.00 s. If the clock is not wound, the pendulums amplitude begins to decay at a rate of 0.53% each pendulum period. a. What is the time constant of...
-
The motion of a nightingales wingtips can be modeled as simple harmonic motion. In one study, the tips of a birds wings were found to move up and down with an amplitude of 8.8 cm and a period of 0.82...
-
A block with a mass of 0.28 kg is attached to a horizontal spring. The block is pulled back from its equilibrium position until the spring exerts a force of 1.0 N on the block. When the block is...

Study smarter with the SolutionInn App



