Calculate the molar translational energy of one molecule of nitrogen and oxygen gas at 0 C...
Fantastic news! We've Found the answer you've been seeking!
Question:
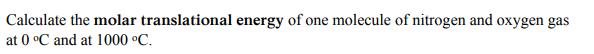
Transcribed Image Text:
Calculate the molar translational energy of one molecule of nitrogen and oxygen gas at 0 C and at 1000 C. Calculate the molar translational energy of one molecule of nitrogen and oxygen gas at 0 C and at 1000 C.
Expert Answer:
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Posted Date:
Students also viewed these chemistry questions
-
(Multiple choice) (1) If the temperature of an ideal gas is doubled while maintaining constant pressure, the average speed of the molecules (a) Remains constant. (b) Increases by a factor of 4. (c)...
-
Carbon monoxide is a gas at 0 C and a pressure of 1.01 10 5 Pa. It is a diatomic gas, each of its molecules consisting of one carbon atom (atomic mass = 12.0 u) and one oxygen atom (atomic mass =...
-
The volume of oxygen gas at 0 C and 104 kPa adsorbed on the surface of 1.00 g of a sample of silica at 0 C was 0.286 cm 3 at 145.4Torr and 1.443 cm 3 at 760Torr. What is the value of V mon ?
-
1. If a firm has already paid an expense or is obligated to pay one in the future, regardless of whether a particular project is undertaken, that expense is a A. Committed cost B. Complementary cost...
-
Show that in choosing the so-called best subset model from a series of candidate models, if the model is chosen that has the smallest s2, this is equivalent to choosing the model with the smallest...
-
As the object in Figure 33.31 is moved closer to the lens, does the size of the image increase, decrease, or stay the same? Figure 33.31 (a) When an object is located between the lens and the focus,...
-
On September 11, 2001, two American Airlines aircraft were hijacked and destroyed in terrorist attacks on The World Trade Center in New York City and the Pentagon in northern Virginia. As a...
-
Lance Brothers Enterprises acquired $720,000 of 3% bonds, dated July 1, on July 1, 2011, as a long-term investment. Management has the positive intent and ability to hold the bonds until maturity....
-
20. Lotsa Lenses paid a dividend of $1.17 last year, and plans a dividend growth rate of 3.40% indefinitely. Lotsa's stock price is now $13.94. What return can Lotsa Lenses' investors expect on their...
-
Consider the calculation of roots of an equation z N = where N 1 is an integer and = ||e j a nonzero complex numb (a) First verify that there are exactly N roots for this equation and that they...
-
what extent do societal attitudes and cultural norms perpetuate stereotypes and stigmatization associated with certain social classes, and how does this impact social cohesion and solidarity?
-
with a Consider a Tank used in certain hydrodynamic experiments. After one experiment, the Tank contains 200L of a dye solution concentration of I g/L. To prepare for the next experiment, The Tank is...
-
Todd wants to start his own business and is debating between organizing the business as a sole proprietorship or a corporation. Explain the pros and cons of both forms of business organization.
-
5. Factor 3x - 14x+8
-
Question Two a. Using appropriate diagrams and reasonable explanation, distinguish between fixed and managed floating exchange rate regimes. b. Differentiate between certainty and uncertainty in...
-
An engineering firm spent $170,000 to purchase and install new computer equipment in its office. The computer equipment has a usable life of 6 years with a salvage value of $820. The company intends...
-
A model car pierces a small compressed gas cylinder to accelerate at a constant rate over a distance of 20 meters. It races against a model car that is propelled by a larger gas cylinder, which...
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
A physics student runs a 1200-W electric heater constantly in her basement bedroom during the winter time. If electric energy costs 9 cents per kilowatt-hour, how much does this electric heating cost...
-
A box of mass m rests on a horizontal table. The coefficient of static friction is s . A force F is applied at an angle as shown in Problem 5-32. (a) Find the force F needed to move the box as a...
-
The Clausius equation of state is P(V bn) = nRT, where b is a constant. Show that the efficiency of a Carnot cycle is the same for a gas that obeys this equation of state as it is for one that obeys...
-
OBrien Corporation is a midsize, privately owned, industrial instrument man ufacturer supplying precision equipment to manufacturers in the Midwest. The corpo ration is 10 years old and operates a...
-
Parktown Medical Center, Inc. is a small health care provider owned by a publicly held corporation. It employs seven salaried physicians, ten nurses, three support staff, and three clerical workers....
-
Figure 10-22 depicts the activities performed in the revenue cycle by the Newton Hardware Company. Required 1 Identify the weaknesses in Newton Hardwares revenue cycle. Explain the resulting threat...

Study smarter with the SolutionInn App



