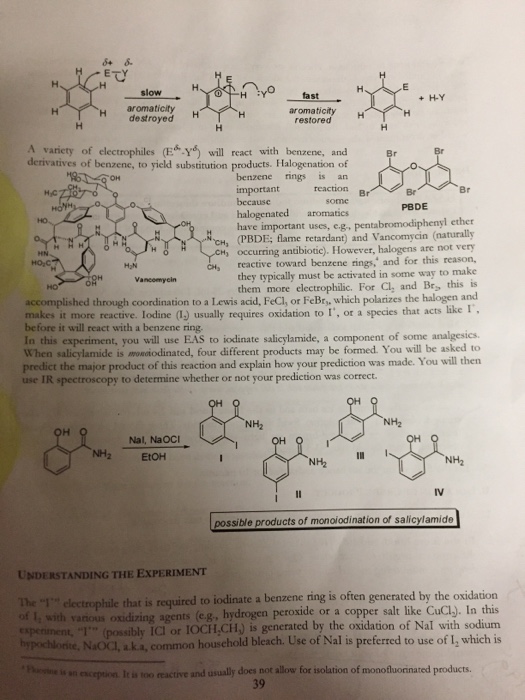
X H H H HCZO HONH HO HN. HOC H A variety of electrophiles (EY) will...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
X² H H H H₂CZO HONH HO HN. HO₂C H A variety of electrophiles (EY) will react with benzene, and derivatives of benzene, to yield substitution products. Halogenation of OH benzene rings is an reaction important because H 84 8 O HH OH slow aromaticity destroyed 8% NH₂ H₂N H. THóẬ H some PBDE halogenated aromatics. CH₂ CH₂ have important uses, e.g., pentabromodiphenyl ether (PBDE; flame retardant) and Vancomycin (naturally CH, occurring antibiotic). However, halogens are not very reactive toward benzene rings, and for this reason, they typically must be activated in some way to make them more electrophilic. For Cl, and Br, this is accomplished through coordination to a Lewis acid, FeCl, or FeBr,, which polarizes the halogen and makes it more reactive. Iodine (1) usually requires oxidation to I, or a species that acts like I , before it will react with a benzene ring. Vancomycin HO Nal, NaOCI EtOH fast aromaticity restored NH₂ он H H NH₂ Br In this experiment, you will use EAS to iodinate salicylamide, a component of some analgesics. When salicylamide is moniodinated, four different products may be formed. You will be asked to predict the major product of this reaction and explain how your prediction was made. You will then use IR spectroscopy to determine whether or not your prediction was correct. OH OH H Br I H + H-Y NH₂ OH Br IV NH₂ 11 possible products of monoiodination of salicylamide UNDERSTANDING THE EXPERIMENT The 1 electrophile that is required to iodinate a benzene ring is often generated by the oxidation of I, with various oxidizing agents (e.g., hydrogen peroxide or a copper salt like CuCl). In this experiment, I (possibly ICI or IOCH,CH) is generated by the oxidation of Nal with sodium hypochlorite, NaOCI, a.k.a, common household bleach. Use of Nal is preferred to use of I, which is Plaine is an exception. It is too reactive and usually does not allow for isolation of monofluorinated products. 39 76.669 volatile and toxic by inhalation. Phenols are very reactive toward EAS and so the reaction mixture must be cooled in an ice bath prior to the addition of NaOCI to slow down the reaction and increase the selectivity for the modinated product. Despite cooling, the reaction is still quite fast as indicated by the quick color changes observed upon addition of NaOCL. Following complete reaction, sodium thiosulfate is added to react with or quench any unreacted I electrophiles, reducing them back to Nal. Because this reaction is done in a basic medium in which the phenol would be deprotonated, the reaction mixture must be acidified prior to product isolation. INSTRUCTIONS HAZARDS: Salicylamide, sodium iodide and sodium thiosulfate are irritants. Sodium hypochlorite and hydrochloric acid are irritants and corrosive. Always wear gloves and appropriate eye protection when handling these compounds. Ethanol is flammable - NO OPEN FLAMES. A. SETTING UP THE EXPERIMENT solven 1. Mass out 0.25 g salicylamide and transfer it to a dry 25 ml. Erlenmeyer flask. Add 5 ml. absolate ethanol and dissolve the salicylamide by swirling the flask. You can warm the flask with your hands to help dissolve the salicylamide. 2. Mass out 0.30 g sodium iodide and add this to your solution of salicylamide. Swirl the flask to dissolve. 3. Once everything is dissolved in the ethanol, CLAMP your flask and cool your mixture to 0 °C in an ice bath for 5-7 minutes. Do not leave the flask in the ice bath for more than 10 minutes as some of the dissolved reagents may begin to precipitate out. If this happens, warm up the flask with your hands. Remove the flask from the ice bath and in the fume hood add 2.4 ml. 6% (w/v) NaOCI (ultra strength bleach) to the solution. Swirl the reaction mixture. There should be a colour change dark red-brown → light yellow. Swirl until the light yellow color is observed, which should take -3-5 minutes. from colourless 5. Let the flask sit on the lab bench andisturbed for 10 minutes. B. ISOLATION OF CRUDE PRODUCT 1. Add 2.5 ml. 10% sodium thiosulfate solution and swirl to mix thoroughly. 4. 2. In the fume hood, add 10% HCI solution drepaise, swirling the flask after every 2-3 drops, until the product begins to precipitate. Once precipitate is observed, continue adding 10% HCI dropwise, monitoring the pH closely using litmus paper, until the mixture becorags acidic. 20 drops 3. Set up a vacuum filtration apparatus and subject the reaction mixture to filtration to collect the crude product. Filter slowly. Allow the crude product to sit under vacuum for 5 minutes to dry. Record the mass of a 100 ml. beaker then transfer your crude product to it and reweigh to determine the mass of your crude product. 4. 59.40 S 58.149. 40 57.49-58.149 = 0.669 mass of crude product = 0. mass of 0.66 .66g recrystallized product = 0.20g C. RECRYSTALLIZATION OF CRUDE PRODUCT 1. Set up a hot water bath. Add 5 mL. 95% ethanol and a stir bar to the beaker containing the crude product. Clamp the beaker in the hot water bath. Heat the solution with stirring until all solid has dissolved. Carefully monitor the reaction as the ethanol can begin boiling rigorously and start bumping 2. Once the solid is dissolved, remove the beaker from the hot water bath and allow it to cool to room temperature. Clamp the beaker in an ice bath to further precipitate crystals. You may scratch the bottom of the beaker with a glass stir rod to promote the formation of crystals if none have started to form after 10 minutes in an ice bath. 70-199 0.209 3. Set up another vacuum filtration apparatus and preweigh the filter papar. Decide which chemical to wet the filter paper and rinse crystals. Collect your recrystallized product. Allow it to dry under vacuum for 5 min. Determine the mass of your recrystallized product. Dispose of all marte materials in the appropriate containers and clean all glassware thoroughly. X² H H H H₂CZO HONH HO HN. HO₂C H A variety of electrophiles (EY) will react with benzene, and derivatives of benzene, to yield substitution products. Halogenation of OH benzene rings is an reaction important because H 84 8 O HH OH slow aromaticity destroyed 8% NH₂ H₂N H. THóẬ H some PBDE halogenated aromatics. CH₂ CH₂ have important uses, e.g., pentabromodiphenyl ether (PBDE; flame retardant) and Vancomycin (naturally CH, occurring antibiotic). However, halogens are not very reactive toward benzene rings, and for this reason, they typically must be activated in some way to make them more electrophilic. For Cl, and Br, this is accomplished through coordination to a Lewis acid, FeCl, or FeBr,, which polarizes the halogen and makes it more reactive. Iodine (1) usually requires oxidation to I, or a species that acts like I , before it will react with a benzene ring. Vancomycin HO Nal, NaOCI EtOH fast aromaticity restored NH₂ он H H NH₂ Br In this experiment, you will use EAS to iodinate salicylamide, a component of some analgesics. When salicylamide is moniodinated, four different products may be formed. You will be asked to predict the major product of this reaction and explain how your prediction was made. You will then use IR spectroscopy to determine whether or not your prediction was correct. OH OH H Br I H + H-Y NH₂ OH Br IV NH₂ 11 possible products of monoiodination of salicylamide UNDERSTANDING THE EXPERIMENT The 1 electrophile that is required to iodinate a benzene ring is often generated by the oxidation of I, with various oxidizing agents (e.g., hydrogen peroxide or a copper salt like CuCl). In this experiment, I (possibly ICI or IOCH,CH) is generated by the oxidation of Nal with sodium hypochlorite, NaOCI, a.k.a, common household bleach. Use of Nal is preferred to use of I, which is Plaine is an exception. It is too reactive and usually does not allow for isolation of monofluorinated products. 39 76.669 volatile and toxic by inhalation. Phenols are very reactive toward EAS and so the reaction mixture must be cooled in an ice bath prior to the addition of NaOCI to slow down the reaction and increase the selectivity for the modinated product. Despite cooling, the reaction is still quite fast as indicated by the quick color changes observed upon addition of NaOCL. Following complete reaction, sodium thiosulfate is added to react with or quench any unreacted I electrophiles, reducing them back to Nal. Because this reaction is done in a basic medium in which the phenol would be deprotonated, the reaction mixture must be acidified prior to product isolation. INSTRUCTIONS HAZARDS: Salicylamide, sodium iodide and sodium thiosulfate are irritants. Sodium hypochlorite and hydrochloric acid are irritants and corrosive. Always wear gloves and appropriate eye protection when handling these compounds. Ethanol is flammable - NO OPEN FLAMES. A. SETTING UP THE EXPERIMENT solven 1. Mass out 0.25 g salicylamide and transfer it to a dry 25 ml. Erlenmeyer flask. Add 5 ml. absolate ethanol and dissolve the salicylamide by swirling the flask. You can warm the flask with your hands to help dissolve the salicylamide. 2. Mass out 0.30 g sodium iodide and add this to your solution of salicylamide. Swirl the flask to dissolve. 3. Once everything is dissolved in the ethanol, CLAMP your flask and cool your mixture to 0 °C in an ice bath for 5-7 minutes. Do not leave the flask in the ice bath for more than 10 minutes as some of the dissolved reagents may begin to precipitate out. If this happens, warm up the flask with your hands. Remove the flask from the ice bath and in the fume hood add 2.4 ml. 6% (w/v) NaOCI (ultra strength bleach) to the solution. Swirl the reaction mixture. There should be a colour change dark red-brown → light yellow. Swirl until the light yellow color is observed, which should take -3-5 minutes. from colourless 5. Let the flask sit on the lab bench andisturbed for 10 minutes. B. ISOLATION OF CRUDE PRODUCT 1. Add 2.5 ml. 10% sodium thiosulfate solution and swirl to mix thoroughly. 4. 2. In the fume hood, add 10% HCI solution drepaise, swirling the flask after every 2-3 drops, until the product begins to precipitate. Once precipitate is observed, continue adding 10% HCI dropwise, monitoring the pH closely using litmus paper, until the mixture becorags acidic. 20 drops 3. Set up a vacuum filtration apparatus and subject the reaction mixture to filtration to collect the crude product. Filter slowly. Allow the crude product to sit under vacuum for 5 minutes to dry. Record the mass of a 100 ml. beaker then transfer your crude product to it and reweigh to determine the mass of your crude product. 4. 59.40 S 58.149. 40 57.49-58.149 = 0.669 mass of crude product = 0. mass of 0.66 .66g recrystallized product = 0.20g C. RECRYSTALLIZATION OF CRUDE PRODUCT 1. Set up a hot water bath. Add 5 mL. 95% ethanol and a stir bar to the beaker containing the crude product. Clamp the beaker in the hot water bath. Heat the solution with stirring until all solid has dissolved. Carefully monitor the reaction as the ethanol can begin boiling rigorously and start bumping 2. Once the solid is dissolved, remove the beaker from the hot water bath and allow it to cool to room temperature. Clamp the beaker in an ice bath to further precipitate crystals. You may scratch the bottom of the beaker with a glass stir rod to promote the formation of crystals if none have started to form after 10 minutes in an ice bath. 70-199 0.209 3. Set up another vacuum filtration apparatus and preweigh the filter papar. Decide which chemical to wet the filter paper and rinse crystals. Collect your recrystallized product. Allow it to dry under vacuum for 5 min. Determine the mass of your recrystallized product. Dispose of all marte materials in the appropriate containers and clean all glassware thoroughly.
Expert Answer:
Answer rating: 100% (QA)
The iodination of salicylamide is an important experiment in the field of organic chemistry The reac... View the full answer

Related Book For 

Fundamental Managerial Accounting Concepts
ISBN: 978-0078025655
7th edition
Authors: Thomas Edmonds, Christopher Edmonds, Bor Yi Tsay, Philip Old
Posted Date:
Students also viewed these accounting questions
-
Which compound is more reactive toward electrophilic substitution (for example, nitration)? OCH or b. CH,CH3 a. ETor
-
Which compound will undergo an electrophilic aromatic substitution reaction more rapidly, benzene or hexadeuteriobenzene? or D- H.
-
Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a Lewis acid such as a. What are the major and minor products of the following reaction? b. Would...
-
Solve the polynomial inequality. (a) Symbolically and (b) Graphically. Use interval notation to write the solution set. x-x>0
-
Why are gains and losses usually shown separately from revenues and expenses from ordinary business activities? What are the implications to the users of the financial statements if gains and losses...
-
Situations in which audit teams are unable to obtain sufficient appropriate evidence necessary to support their opinion on the entity's financial statements are referred to as scope limitations....
-
Outline how standard desktop applications such as a spreadsheet or database can be used to set up a basic T&D system.
-
Allen Abbott has a wide-curving, uphill driveway leading to his garage. When there is a heavy snow, Allen hires a local carpenter, who shovels snow on the side in the winter, to shovel his driveway....
-
How does the dynamic interplay between protgs and mentors within a mentorship framework contribute to the cultivation of cognitive dexterity and strategic foresight in navigating complex professional...
-
1. Marketing analysts use market position maps to display visually the customers perceptions of a firm in relation to its competitors regarding two attributes. Prepare a market position map for Alamo...
-
Required: Your submission for assignments 1 and 3 should be in the format below: Potential risk-description General and industry-specific economic trends and conditions Competitive environment...
-
What is needed how much is needed and when it is needed in materials management?
-
What management feature provides advanced document control solutions?
-
What main piece of information does the aging report provide? A. Demographic information on each patient. B. Payments received from third-party payers. C. Unpaid balances in each patient account. D....
-
Research a company's annual report. Prepare a video summary of the firm's financial ratios and financial statements. During the video summary, annotate the annual report and share relevant...
-
Evelyn and Carter have been married for 46 years. They are in their retirement and have 2 kids that are financially independent.They have five grandchildren, all under the age of 10. Carter is in...
-
In a limited partnership, the limited partner: a) cannot actively participate in the management of the partnership. b) can only invest their money in the company for a limited time period. c) cannot...
-
How does health insurance risk differ from other types of insurance risk (e.g., automobile or homeowners insurance)? What is the difference between cost sharing and cost shifting? Is retiree health...
-
Daniel Singh, the controller of Meier Corporation, is trying to prepare a sales budget for the coming year. The income statements for the last four quarters follow. Historically, cost of goods sold...
-
Dunlop Corporation is about to start a business as an agricultural products distributor. Because its customers will all be retailers, Dunlop will sell its products solely on account. The company...
-
During 2014, Welch Manufacturing Company incurred $67,000,000 of research and development (R&D) costs to create a long-life battery to use in computers. In accordance with FASB standards, the entire...
-
Has the U.S. economy experienced inflation or deflation during recent recessions? Explain.
-
How do you think recessions influence elections?
-
Inflation is soaring and employment is beginning to show sustained improvement. The unemployment rate is 6 percent, compared to its 3.5 percent rate prepandemic. The CPI grew 5.4 percent in June, and...

Study smarter with the SolutionInn App


