Calculate the solubility at 25 C of PbCrO, in pure water and in a 0.0160 M...
Fantastic news! We've Found the answer you've been seeking!
Question:
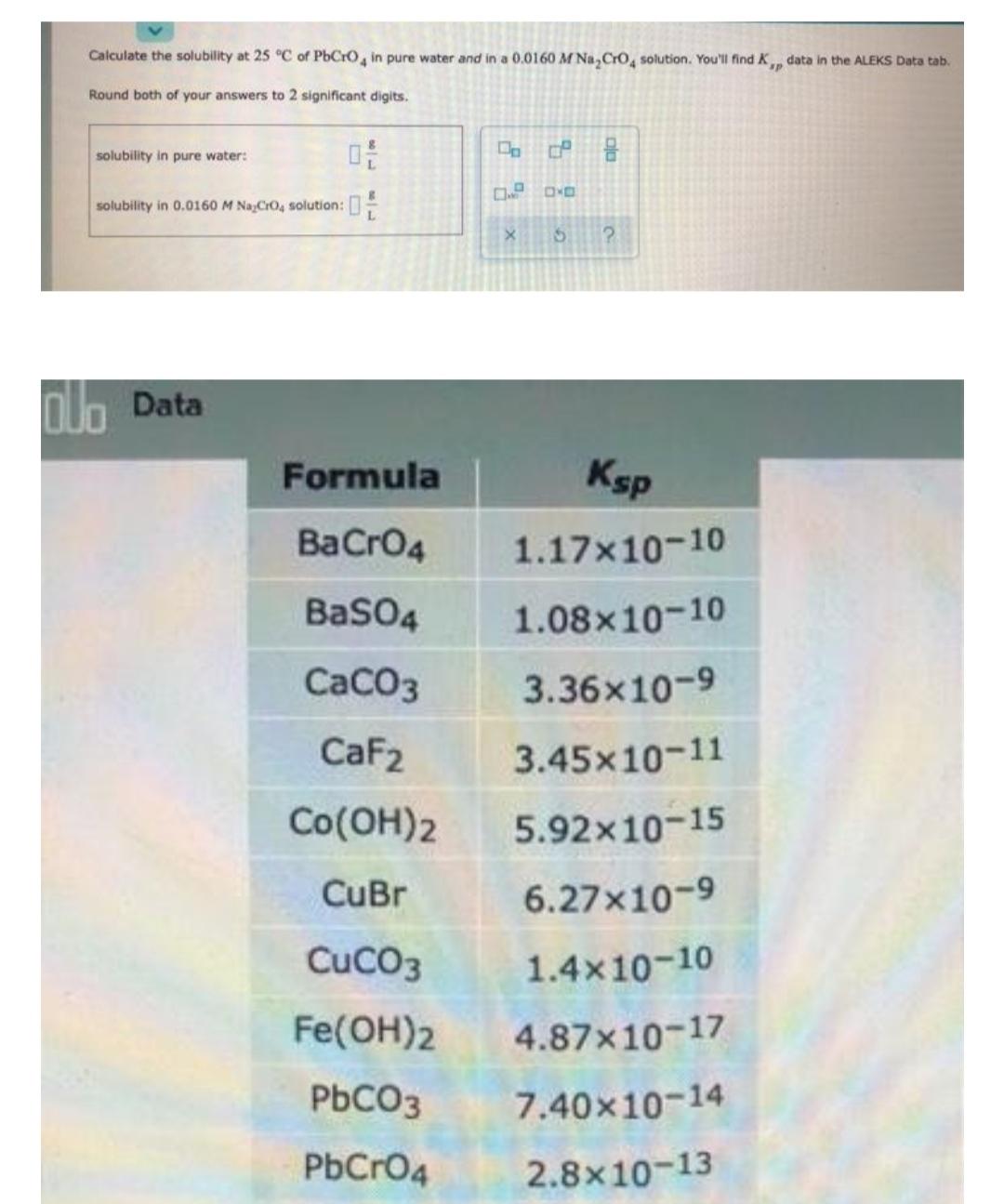

Transcribed Image Text:
Calculate the solubility at 25 °C of PbCrO, in pure water and in a 0.0160 M Na, Cro, solution. You'll find K data in the ALEKS Data tab. Round both of your answers to 2 significant digits. solubility in pure water: D OO solubility in 0.0160 M NaCrO4 solution:: olo Data Formula Ksp BaCrO4 1.17x10-10 BasO4 1.08x10-10 CaCO3 3.36x10-9 CaF2 3.45x10-11 Co(OH)2 5.92x10-15 CuBr 6.27x10-9 CuCO3 1.4x10-10 Fe(OH)2 4.87x10-17 PBCO3 7.40x10-14 PbCrO4 2.8x10-13 Co(OH)2 5.92x10-15 CuBr 6.27x10-9 CuCO3 1.4x10-10 Fe(OH)2 4.87x10-17 PBCO3 7.40x10-14 PbCrO4 2.8x10-13 PBF2 3.3x10-8 Mg(OH)2 5.61x10-12 Ni(OH)2 5.48x10-16 AgBrO3 5.38x10-5 Ag2CO3 8.46x10-12 AgCI 1.77x10-10 Ag2CrO4 1.12x10-12 SrCO3 5.60x10-10 ZnCO3 1.46x10-10 Zn(OH)2 3.0x10-17 AgBr 5.35x10-13 AuCI 1.77x10-10 Calculate the solubility at 25 °C of PbCrO, in pure water and in a 0.0160 M Na, Cro, solution. You'll find K data in the ALEKS Data tab. Round both of your answers to 2 significant digits. solubility in pure water: D OO solubility in 0.0160 M NaCrO4 solution:: olo Data Formula Ksp BaCrO4 1.17x10-10 BasO4 1.08x10-10 CaCO3 3.36x10-9 CaF2 3.45x10-11 Co(OH)2 5.92x10-15 CuBr 6.27x10-9 CuCO3 1.4x10-10 Fe(OH)2 4.87x10-17 PBCO3 7.40x10-14 PbCrO4 2.8x10-13 Co(OH)2 5.92x10-15 CuBr 6.27x10-9 CuCO3 1.4x10-10 Fe(OH)2 4.87x10-17 PBCO3 7.40x10-14 PbCrO4 2.8x10-13 PBF2 3.3x10-8 Mg(OH)2 5.61x10-12 Ni(OH)2 5.48x10-16 AgBrO3 5.38x10-5 Ag2CO3 8.46x10-12 AgCI 1.77x10-10 Ag2CrO4 1.12x10-12 SrCO3 5.60x10-10 ZnCO3 1.46x10-10 Zn(OH)2 3.0x10-17 AgBr 5.35x10-13 AuCI 1.77x10-10
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The pH at 25 C of an aqueous solution of the sodium salt of -hydroxyquinoline (NaC10H7O) is 11.12. Calculate the concentration of C10H7O- in this solution, in moles per liter. Ka for HC10H7O is equal...
-
What is the pH at 25 C of water saturated with CO 2 at a partial pressure of 1.10 atm? The Henry's law constant for CO 2 at 25 C is 3.1 10 2 mol/L-atm. The CO 2 is an acidic oxide, reading with H 2...
-
Calculate the solubility of Ag,CO3 (a) in pure water and (b) in a solution in which [CO,]= 0.203 M. %3D Solubility in pure water = M Solubility in 0.203 M Co,2 - M %3!
-
Cooper Movie Studio Corp. makes movies and is interested in lowering its operating costs for the following year, while maintaining the high quality and appeal of its movies. Cooper's management is...
-
Select a favourite Internet site for a brand and investigate how it achieves the objectives outlined in this chapter.
-
According to the rule adopted by the majority of courts to determine accountants liability to third parties, could Chase be liable to Superior? Superior Wholesale Corporation planned to purchase...
-
What should we do if there is more than one primary actor for the use case?
-
Variable price and usage variances and fixed manufacturing overhead cost variances Kemp Tables, Inc. (KTI), makes picnic tables of 2 3 4 planks of treated pine. It sells the tables to large retail...
-
Midnight Sun Apparel Company uses normal costing, and manufacturing overhead is applied to work-in-process on the basis of machine hours. On January 1 of the current year, there were no balances in...
-
The following data relates to Prism traders limited for the year ended 31st December, 2018. Shs Cash and marketable securities Fixed Assets Sales Net Income Quick Ratio Current Ratio Average...
-
Breanne is going away to university. She has saved up some money from part-time jobs and plans to buy or lease a new vehicle. She has found a compact car that she likes and is trying to decide...
-
Which of these are functional interfaces? A. Lion B. Tiger C. Both Lion and Tiger D. Neither is a functional interface. E. The code does not compile. interface } Lion { public void roar (); default...
-
Which variable declaration is the first line not to compile? A. b3 B. h2 C. b4 D. h3 E. All of the lines compile. } public class Complex { class Building {} class House extends Building {} public...
-
What is the output of the following application? A. 5 B. 6 C. 8 D. The code does not compile. package stocks; public class Bond { } private static int price = 5; public boolean sell() { if (price...
-
Fill in the blanks: ___________________ methods always have the same name but a different list of parameters, while ___________________ methods always have the same name and the same return type. A....
-
Which methods compile? A. Just getNumRakes() B. Just getNumShovels() C. Both methods D. Neither method private static int numShovels; private int numRakes; public int getNumShovels () { return...
-
Your manager believes that intergroup conflict can encourage healthy competition in your department so he decides to give the team with the lowest amount of product defects a trip to Hawaii. What...
-
Calculate Total Contribution Margin for the same items. Total Revenue Total Variable Costs Total Contribution Margin $50.00 a. $116.00 $329.70 b. $275.00 $14,796.00 $7,440.00 c. $40,931.25 d....
-
An unknown salt is either NaF,NaCl, or NaOCl. When 0.050 mol of the salt is dissolved in water to form 0.500 L of solution, the pH of the solution is 8.08.What is the identity of the salt?
-
Calculate the percentage by mass of oxygen in the following compounds: (a) Morphine, C17H19NO3 (b) Codeine, C18H21NO3 (c) Cocaine, C17H21NO4 (d) Tetracycline, C22H24N2O8 (e) Digitoxin, C41H64O13 (f)...
-
Describe how a cholesteric liquid crystal phase differs from a nematic phase?
-
See the option quote on IBM from the CBOE Web site on the next page showing options expiring in March and April 2022. a. Which option contract had the most trades that day? b. Which option contract...
-
Two European call options with a strike price of \($50\) are written on two different stocks. Suppose that tomorrow, the low-volatility stock will have a price of \($50\) for certain. The...
-
It is February 21, 2022, and you have decided to purchase 10 June call contracts on eBays stock with an exercise price of \($57.50.\) Because you are buying, you must pay the ask price. How much...

Study smarter with the SolutionInn App


