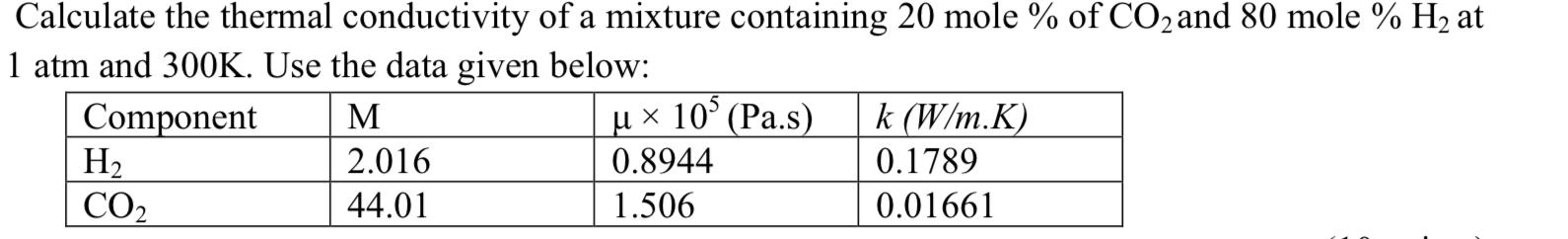
Calculate the thermal conductivity of a mixture containing 20 mole % of CO2 and 80 mole...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculate the thermal conductivity of a mixture containing 20 mole % of CO2 and 80 mole % H2 at 1 atm and 300K. Use the data given below: Component H2 CO2 µ x 10° (Pa.s) 0.8944 M k (W/m.K) 2.016 0.1789 44.01 1.506 0.01661 Calculate the thermal conductivity of a mixture containing 20 mole % of CO2 and 80 mole % H2 at 1 atm and 300K. Use the data given below: Component H2 CO2 µ x 10° (Pa.s) 0.8944 M k (W/m.K) 2.016 0.1789 44.01 1.506 0.01661
Expert Answer:
Answer rating: 100% (QA)
ook 1 14 2 das Llit Mx J8 M daz 1 V4 124401 108944 12 1 ... View the full answer

Related Book For 

Principles of heat transfer
ISBN: 978-0495667704
7th Edition
Authors: Frank Kreith, Raj M. Manglik, Mark S. Bohn
Posted Date:
Students also viewed these accounting questions
-
The thermal conductivity of a material may be determined in the following manner. Saturated steam 2.41 105 N/m2 is condensed at the rate of 0.68 kg/h inside a hollow iron sphere that is 1.3 cm thick...
-
The thermal conductivity of a refrigerator door is to be determined by measuring the surface temperatures and heat flux when steady operating conditions are reached. A = 0.03 m2, QUOTE
-
Calculate the thermal conductivity of nitrogen (C; m = 20.8 J K-1 mol-1, a= 0.43 nm3) at room temperature (20C).
-
Table illustrates the quantities, marginal costs, average variable costs, and average costs of a competitive firm. Refer to table 4. How much is the unit profit at price =$30? [the problem is based...
-
Suppose you are designing a system to summarize information from rsums submitted to a companys human resources department. Create a list of the information that you think would be useful to collect...
-
Kartman Corporation is evaluating four real estate investments. Management plans to buy the properties today and sell them three years from today. The annual discount rate for these investments is...
-
In testing prepaid insurance, how does an auditor address rights and obligations?
-
The Ashton Furniture Company manufactures coffee tables and chest of drawers. Last year the companys cost of goods sold was $3,700,000, and it carried inventory of oak, pine, stains, joiners, and...
-
Create and Design a form as given below: (5 Marks) Land Of Boxes Order Form Pomode 200 Credit Card Details Cad Part Neer Quantity Unit Price tal F 310 13500 305 2400 320 330 Cadabr Delivery Address p...
-
Required: Nancy, who is 59 years old, is the beneficiary of a $220,000 life insurance policy. What amount of the insurance proceeds is taxable under each of the following scenarios? (Do not round any...
-
The accelerating voltage is Vacc = 2 3 2 V The deflecting voltage is Vdef = 2 2 V The length L of the deflecting plates is 1 . 9 9 cm The separation d between the deflecting plates is 4 mm The...
-
Many of you have worked for or experienced various types of leaders. Identify one major quality you believe makes a good leader and explain how this quality interrelates to one values? Please explain...
-
Accountants, Ethics, and the Financial Statements ( 2 5 Marks ) For some time, Hernadez Pty Co . a beverage manufacturer has been selling to the west coast region, however recently their market share...
-
Rose Apothecary Inc. Issued a 3 - year $ 1 0 0 , 0 0 0 bond with an 8 % stated rate sold to yield 1 0 % on January 1 , 2 0 2 4 . Interest is payable annually on December 3 1 st . Bond Issue Costs are...
-
Ralph contributes land with a basis and value of $ 1 2 0 , 0 0 0 in exchange for a 3 0 % share of the profits and losses in Orange LLC . Orange is a calendar year LLC . Orange generates $ 3 0 0 , 0 0...
-
Tax on purchases during the year Interest from a savings account Interest from City of Boston Bonds Dividend from 3 M stock $ 1 , 4 0 0 5 0 0 9 , 0 0 0 2 , 6 0 0 1 , 0 0 0 6 , 2 0 0 8 0 0 7 0 0 4 0 0...
-
Refer to IRS 2022 Publication 15-T to answer the following questions: Cheyenne is paid $10,009.54 monthly as a regional manager for Really Ritzy Restaurants. She is married filing jointly, according...
-
Conduct a VRIO analysis by ranking Husson University (in Maine) business school in terms of the following six dimensions relative to the top three rival schools. If you were the dean with a limited...
-
A horizontal 3-mm-thick flat copper plate, 1 m long and 0.5 m wide, is exposed in air at 27?C to radiation from the sun. If the total rate of solar radiation absorbed is 300 W and the combined...
-
A 0.15 m square circuit board is to be cooled in a vertical position as shown. The board is insulated on one side while on the other, 100 closely spaced square chips are mounted, each of which...
-
A large slab of steel 0.1 m thick has in it a 0.1 m-diam hole, with axis normal to the surface. Considering the sides of the hole to be black, specify the rate of radiative heat loss from the hole in...
-
Calculate the volume occupied by isopropanol vapour at \(200^{\circ} \mathrm{C}\) and 10 bar by using (a) Ideal gas equation of state (b) Virial equation of state (c) Virial equation of state \[...
-
The Dieterici equation of state is given by \[ P(V-b) \exp \left(\frac{a}{R T V} ight)=R T \] where \(a\) and \(b\) are constants. Develop the relations to determine the constants \(a\) and \(b\) in...
-
At \(17^{\circ} \mathrm{C}\), at constant pressure, the heat of combustion of amorphous carbon is 96960 cal and that of \(\mathrm{CO}\) to \(\mathrm{CO}_{2}\) is \(67960 \mathrm{cal}\). Determine the...

Study smarter with the SolutionInn App


