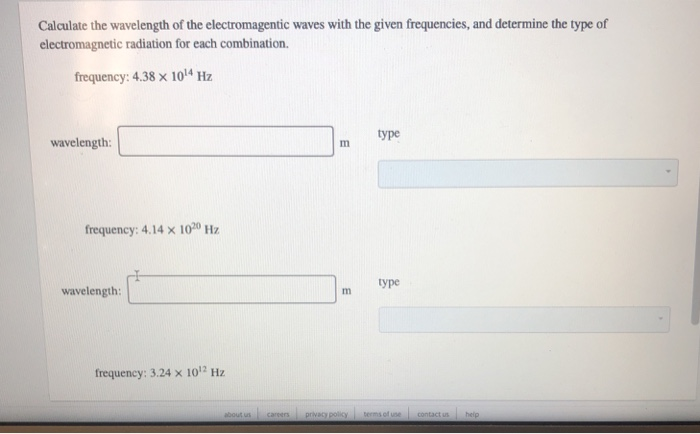
Calculate the wavelength of the electromagentic waves with the given frequencies, and determine the type of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
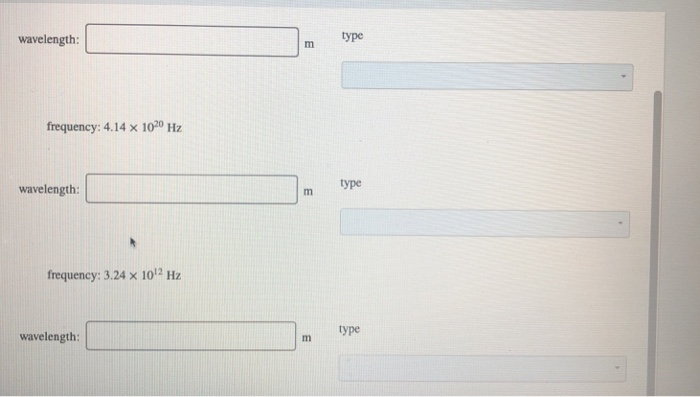
Calculate the wavelength of the electromagentic waves with the given frequencies, and determine the type of electromagnetic radiation for each combination. frequency: 4.38 x 10¹4 Hz wavelength: frequency: 4.14 x 1020 Hz wavelength: frequency: 3.24 x 10¹2 Hz about us careers m E privacy policy type type terms of use contact us help wavelength: frequency: 4.14 x 1020 Hz wavelength: frequency: 3.24 x 10¹2 Hz wavelength: m m m type type type Calculate the wavelength of the electromagentic waves with the given frequencies, and determine the type of electromagnetic radiation for each combination. frequency: 4.38 x 10¹4 Hz wavelength: frequency: 4.14 x 1020 Hz wavelength: frequency: 3.24 x 10¹2 Hz about us careers m E privacy policy type type terms of use contact us help wavelength: frequency: 4.14 x 1020 Hz wavelength: frequency: 3.24 x 10¹2 Hz wavelength: m m m type type type
Expert Answer:
Answer rating: 100% (QA)
Answer A Frequency f 438 1014 Hz using c f X 3108 4381014 X ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
Calculate the wavelength of a photon needed to excite a transition between neighbouring energy levels of a harmonic oscillator of effective mass equal to that of an oxygen atom (15.9949 u) and force...
-
Calculate the wavelength of a 0.23-kg ball traveling at 0.10m/s.
-
Calculate the wavelength of photons needed to produce NMR transitions in free protons in a 1.000-T field. In what region of the spectrum does it lie?
-
A 0.500-kg block, attached to a spring with length 0.60 m and force constant 40.0 N/m, is at rest with the back of the block at point A on a frictionless, horizontal air table (Fig. 7.44). The mass...
-
Assume that you borrow $25,000 on April 1, 2004, at an annual rate of 7%. How much will you owe on March 31, 2005 if you make no payments until that date? How much will you owe on March 31, 2006 if...
-
Brondon Corp. purchased a put option on Mykia common shares on July 7, 2011, for $480. The put option is for 350 shares, and the strike price is $50. The option expires on January 31, 2012. The...
-
Question: Sun operates an upscale sandwich shop in New Jersey, in a storefront that she leases from Ricky for $18,000 per month. The lease, which expires soon, allows Sun to renew for five years, at...
-
Arantxa Corporation made the following cash purchases of securities during 2008, which is the first year in which Arantxa invested in securities. 1. On January 15, purchased 10,000 shares of Sanchez...
-
which way describes a triangle with one side 5 centimeters, 12 centimeters, and 13 centimeters 2 answers
-
Martha Millon, financial manager for Fish & Chips Inc., has been asked to perform a lease-versus-buy analysis on a new computer system. The computer system has an after-tax cost of $975,000, and if...
-
Question Two (a) A six-month European Call option on a non-dividend paying Stock Index has a strike price of $4900. The index price is $5000, the risk-free rate is 5% per annum, and the value of u...
-
What effect does the higher error rate have on the sample sizes needed to provide reliable sample results in attributes sampling?
-
What is precision?
-
Contrast computer simulation and parallel simulation as used in this chapter.
-
When Maggie Lance took over Air-Techs packaging and shipping (P & S) department, it was notorious within the company as a dead-end, high-stress, uncomfortable place to work. Internal politics,...
-
What is confidence level?
-
Apex Learning - Test though Josiah wasn't sure it was a good idea, he didn't say anything. The president told all of the members to wear a bag with eye holes cut out to look like bulls eyes. When...
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
You have a concentration cell with Cu electrodes and [Cu2+] = 1.00 M (right side) and 1.0 1024 M (left side). a. Calculate the potential for this cell at 25oC. b. The Cu21 ion reacts with NH3 to...
-
The decomposition of hydrogen peroxide was studied at a particular temperature. The following data were obtained, where Determine the integrated rate law, the differential rate law, and the value of...
-
The carbonate ion (CO32-) can act as either a monodentate or a bidentate ligand. Draw a picture of CO32- coordinating to a metal ion as a bidentate and as a monodentate ligand. The carbonate ion can...
-
On July 1, 2020, West Company purchased for cash, eight \(\$ 10,000\) bonds of North Corporation to yield \(10 \%\). The bonds pay \(9 \%\) interest, payable on a semiannual basis each July 1 and...
-
Repeat Exercise 14-43, assuming discounts and premiums are amortized using the straight-line interest method. Exercise 14-43 On July 1, 2020, West Company purchased for cash, eight \(\$ 10,000\)...
-
On January 1, 2020, Lazer Inc. purchased for cash, ten \(\$ 1,000,4 \%\) bonds of Star Corp. at par. The bond interest is paid annually on January 1 of each year, and the bond maturity date is...

Study smarter with the SolutionInn App


