Threonine, an amino acid, has both an acid group and a basic group in its structure...
Fantastic news! We've Found the answer you've been seeking!
Question:
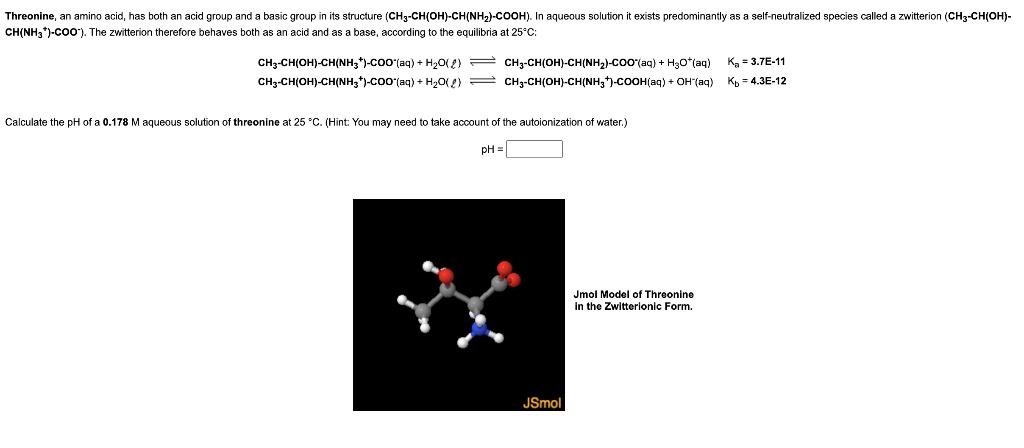
Transcribed Image Text:
Threonine, an amino acid, has both an acid group and a basic group in its structure (CH3-CH(OH)-CH(NH₂)-COOH). In aqueous solution it exists predominantly as a self-neutralized species called a zwitterion (CH3-CH(OH)- CH(NH3*)-COO). The zwitterion therefore behaves both as an acid and as a base, according to the equilibria at 25°C: CH3-CH(OH)-CH(NH3)-COO(aq) + H₂O(2) CH3-CH(OH)-CH(NH3)-COO (aq) + H₂O(2) CH3-CH(OH)-CH(NH,)-COO^(aq) + HyO*(aq) CH3-CH(OH)-CH(NH3)-COOH(aq) + OH (aq) Calculate the pH of a 0.178 M aqueous solution of threonine at 25 °C. (Hint: You may need to take account of the autoionization of water.) pH = JSmol Jmol Model of Threonine in the Zwitterionic Form. K₂ = 3.7E-11 Kb =4.3E-12 Threonine, an amino acid, has both an acid group and a basic group in its structure (CH3-CH(OH)-CH(NH₂)-COOH). In aqueous solution it exists predominantly as a self-neutralized species called a zwitterion (CH3-CH(OH)- CH(NH3*)-COO). The zwitterion therefore behaves both as an acid and as a base, according to the equilibria at 25°C: CH3-CH(OH)-CH(NH3)-COO(aq) + H₂O(2) CH3-CH(OH)-CH(NH3)-COO (aq) + H₂O(2) CH3-CH(OH)-CH(NH,)-COO^(aq) + HyO*(aq) CH3-CH(OH)-CH(NH3)-COOH(aq) + OH (aq) Calculate the pH of a 0.178 M aqueous solution of threonine at 25 °C. (Hint: You may need to take account of the autoionization of water.) pH = JSmol Jmol Model of Threonine in the Zwitterionic Form. K₂ = 3.7E-11 Kb =4.3E-12
Expert Answer:
Answer rating: 100% (QA)
The problem involves calculating the pH of a solution of the amino acid threonine Threonine has both ... View the full answer

Related Book For 

Financial Management for Decision Makers
ISBN: 978-0138011604
2nd Canadian edition
Authors: Peter Atrill, Paul Hurley
Posted Date:
Students also viewed these accounting questions
-
In an effort to make better use of its resources, the New York City Food Bank engaged in lean process improvement. This employee-driven kaizen effort resulted in a new method for packing meals for...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
How would you compare the value of direct definition from friends in face-to-face conversation and strangers comments on a YouTube posting?
-
On July 1, Laura Corporation purchases 500 shares of its $5 par value common stock for the treasury at a cash price of $8 per share. On September 1, it sells 300 shares of the treasury stock for cash...
-
a. How much work is performed when you push a couch of mass 44 kg a distance of 2.5 m using a force of 135 N? b. You perform 556 J of work lifting a box to a height of 1.3 m. How much force did you...
-
Do consumer products and services perform missions?
-
Crabtree Co. purchased $60,000 of 6%, 15-year Thomas County bonds on June 20, 2014, directly from the county, at their face amount plus accrued interest. The bonds pay semiannual interest on May 1...
-
Evaluate 3x2+8x+8, when x = 3.
-
Slopes Inc. manufactures and sells snowboards. Slopes manufactures a single model, the Pipex. In the summer of 2015, Slopes's accountant gathered the following data to prepare budgets for 2016. These...
-
Why do you think people cheat on their taxes? What could the government do to decrease how much/many people cheat on their taxes? Do you think that if people knew exactly where their tax dollars were...
-
Prepare the journal entries for all the part 1 transactions Create ???? accounts for all accounts and add the part 1 transactions Part 1 Transactions: January 1 - Phin invested $ 1 8 , 5 0 0 of his...
-
1 . ABC Corporation manufactures and sells coloring materials. Its selling price per unit is Php 1 5 . The company operated for 2 1 days for the month of January. Its daily wage earners are paid Php...
-
Wardell Company purchased a mini computer on January 1, 2022, at a cost of $40,000. The computer has been depreciated using the straight-line method over an estimated five-year useful life with an...
-
What is the result of this code? ArrayList flowers new ArrayList (); flowers.add("daisy"); flowers.add("rose"); flowers.add("orchid"); System.out.println(flowers.get(1));...
-
Define the following common terms used in coding programs: Interpreter Function I Program Variable List Dictionary print() input() vs output If Statements IDLE
-
Let A = {1,2,3,4,5), B = {3,4,5,6,7), and C= {2,3,5).
-
Is that Yelp review real or fake? The article A Framework for Fake Review Detection in Online Consumer Electronics Retailers (Information Processing and Management 2019: 12341244) tested five...
-
Shares of Exem Radiator Limited (ERL), a government-controlled company, currently sell for $45.00 on the Toronto Stock Exchange. ERL pays an annual dividend of $8.00 per share at year-end. Government...
-
Golden Resources Inc. is predicting the following cash inflows for a proposed new project under three different growth scenarios: Required: (a) Calculate the present value of the cash inflows for...
-
You have gathered the following data for your new line of engagement rings. Sales ........................................... $10,000,000 Fixed costs ....................................8,000,000...
-
A multiple-leaf spring used in automobiles is shown in Fig. 12.17. It consists of five leaves, each of thickness \(t=0.65 \mathrm{~cm}\) and width \(w=3.8 \mathrm{~cm}\). For the multiple-leaf spring...
-
Fill in the Blank. For a thin beam element, __________ degrees of freedom are considered at each node.
-
True or False. The Rayleigh-Ritz method assumes that the solution is a series of functions that satisfy the boundary conditions of the problem.

Study smarter with the SolutionInn App


